INTRODUCTION
Over the last decade, the profile of patients undergoing percutaneous coronary intervention (PCI) has changed considerably. It is increasingly common to treat patients with advanced atherosclerosis, previous coronary revascularization, and more complex coronary anatomy. In addition, these patients often have systemic comorbidities, such as chronic renal dysfunction or diabetes, which increase the complexity of the procedure and raise the risk of associated complications.
In this context, the need to optimize the use of iodinated contrast has become particularly relevant, given that high doses can cause acute renal injury, known as contrast-induced nephropathy (CIN). This condition is associated with higher morbidity and mortality, longer hospital stays, and a significant increase in healthcare costs. Although CIN is a multifactorial phenomenon, the amount of contrast used during PCI has been identified as one of the main risk factors, leading to the search for new strategies to minimize its use without compromising the diagnostic and therapeutic efficacy of the procedure.
To address this issue, tools have been developed to optimize contrast use during PCI. Among them, the Dynamic Coronary Roadmap (DCR) appears to be an innovative solution. This software projects a real-time reference image of the coronary anatomy onto the fluoroscopy. In this way, the DCR generates a dynamic and automated map of the coronary arteries, superimposed on the live fluoroscopy image, allowing devices (guides, balloons, and stents) to be guided more efficiently and safely with less contrast use. The technical feasibility of the DCR has already been evaluated in previous studies. In an analysis of 936 cases, the quality of anatomical superimposition with angiography was considered “fit for use” in 99.5% of cases, with low inter- and intra-observer variability. These findings support the reliability of the technology for integration into clinical practice. (1)
The present study aims to evaluate the impact of DCR use on contrast utilization during PCI at a center of Argentina. Despite international evidence supporting its efficacy, the reported local experience is limited, which justifies the need to generate specific data for our healthcare reality. The findings of this study could facilitate the adoption of this technology in interventional cardiology centers in the country, with direct implications in patient safety, operational efficiency, and resource optimizatio.
METHODS
This single-center, retrospective study analyzed the database of the Endovascular Therapy Service at Instituto Cardiovascular San Gerónimo (Santa Fe, Santa Fe), which has been collecting information since 2017. The database allows for systematic 30-day and annual follow-up, with monitoring of population characteristics and technical and clinical outcomes. Demographic data, cardiovascular history, procedures performed, and clinical evolution are analyzed. Contrast consumption, recorded in the Radiology Report prepared for each patient admitted to the Hemodynamics Room, includes the amount used and radiation doses. In procedures that combine cine coronary angiography and PCI, contrast and radiation consumption are reported separately for each intervention.
Study population
The analysis included consecutive patients undergoing PCI between January and December 2024, who were classified according to whether the procedure was guided by DCR (DCR group) or conventional angiography (control group). In all cases, at least 25 mL of contrast was injected manually. Only cases with cardiogenic shock on admission (systolic blood pressure <100 mmHg, heart rate >100 bpm, poor distal perfusion requiring inotropic drugs or mechanical circulatory support devices) or those in whom only lateral branch lesions were treated without involvement of the main coronary arteries were excluded. No other exclusion criteria were applied.
All procedures were performed by the same team of operators with experience in coronary intervention, in the same catheterization laboratory and using the same angiographic system (Azurion 3 M15, Philips).
Definitions
Complex PCI is defined as that involving the treatment of multiple vessels, unprotected left main coronary artery approach, intervention on a venous bypass graft, management of a true bifurcation, or treatment of lesions with severe calcification requiring calcium ablation techniques or thrombotic lesions. Ad hoc PCI is defined as PCI performed immediately after diagnostic coronary angiography, while planned PCI is performed as a scheduled intervention at a later time after coronary angiography. CIN was defined as an increase of ≥0.5 mg/dL or ≥25% of baseline serum creatinine within 48-72 hours after the procedure.
Outcomes
The primary objective of this study was to evaluate whether the use of DCR reduces the total contrast volume in PCI. The secondary objectives were to analyze the total amount of radiation used by PCI in each group, as well as post-PCI renal function evaluation according to the use or non-use of DCR.
Statistical analysis
Qualitative variables are expressed as percentages and were evaluated using the chi-square test. Quantitative variables were subjected to normality tests (Kolmogorov-Smirnov test or Shapiro-Wilk test, as appropriate) and histogram parameter measurements were used: skewness and kurtosis. Those variables that met the criteria for normality were expressed as mean ± standard deviation (SD), and otherwise as median and interquartile range (IQR).
Baseline demographic, clinical, and procedural characteristics were compared using Student’s t-test or the MannWhitney U test for continuous variables according to their distribution, and the chi-square test or Fisher’s exact test for categorical variables. A two-tailed significance level of 0.05 was set for all analyses.
Differences in the primary outcome (total contrast consumption) as well as in the secondary outcome were evaluated using Student’s t-test or the Mann-Whitney U test depending on their distribution and variance. Linear regression was used to evaluate the impact of DCR use adjusted for confounding variables: age, sex, vessel treated, and PCKI complexity.
RESULTS
This analysis included 480 cases of patients treated with PTCI between January and December 2024, of which 201 were guided by DCR (41.8%) and 279 by angiography (58.2%).
Demographic variables are detailed in Table 1. No significant differences were observed in terms of age and sex, although patients in the DCR group had a higher prevalence of dyslipidemia.
Table 2 reports the procedure data. There were no significant differences between the two groups in terms of type of procedure (ad hoc PCI vs. scheduled PCI) or the vessel treated. There was a significant difference in the proportion of complex PCI in the DCR branch compared with the control group (39.6% vs. 17.6%, p < 0.001).
Table 1
Baseline variables
| Variable | DCR group (n= 201) | Control group (n= 279) | p |
|---|---|---|---|
| Age, years | 69 (62-75) | 68 (60 - 75) | 0.507 |
| Male gender | 78.8 | 75 | 0.819 |
| BMI, kg/m2 | 28 (23 - 31) | 28 (22 - 31) | 0.653 |
| Diabetes mellitus | 23.8 | 21.6 | 0.596 |
| Dyslipidemia | 79.7 | 67.3 | 0.006 |
| HTN | 84.9 | 86.1 | 0.723 |
| Previous PTCA | 32.1 | 30.7 | 0.779 |
| LVEF < 30% | 2 | 2.6 | 0.951 |
| Baseline creatinine, mg/dL | 0.88 (0.78 - 1.0) | 0,90 (0.73 - 1.0) | 0.896 |
BMI: body mass index; DCR: Dynamic Coronary Roadmap; HTN: hypertension; LVEF: left ventricular ejection fraction; PCI: percutaneous coronary intervention;
Continuous variables are presented as median (interquartile range) and categorical variables as percentage.
Table 2
Type of procedure and vessel treated
| Variable | DCR group (n= 201) | Control group (n= 279) | p |
|---|---|---|---|
| Type of procedure | |||
| Ad hoc PCI | 58.1 | 53.1 | 0.306 |
| Scheduled PCI | 41.9 | 46.9 | |
| Treated vessel | |||
| LMCA | 4.1 | 7.6 | 0.195 |
| LAD | 41.9 | 37.1 | |
| Cx | 25.6 | 33.1 | |
| RCA | 25 | 25.7 | |
| Complex PCI | 39.6 | 17.6 | < 0.001 |
Cx: circumflex artery; DCR: Dynamic Coronary Roadmap; LAD: left anterior descending artery; LMCA: left main coronary artery; PCI: percutaneous coronary coronary intervention; RCA: right coronary artery
Categorical variables are presented as percentage.
Contrast consumption
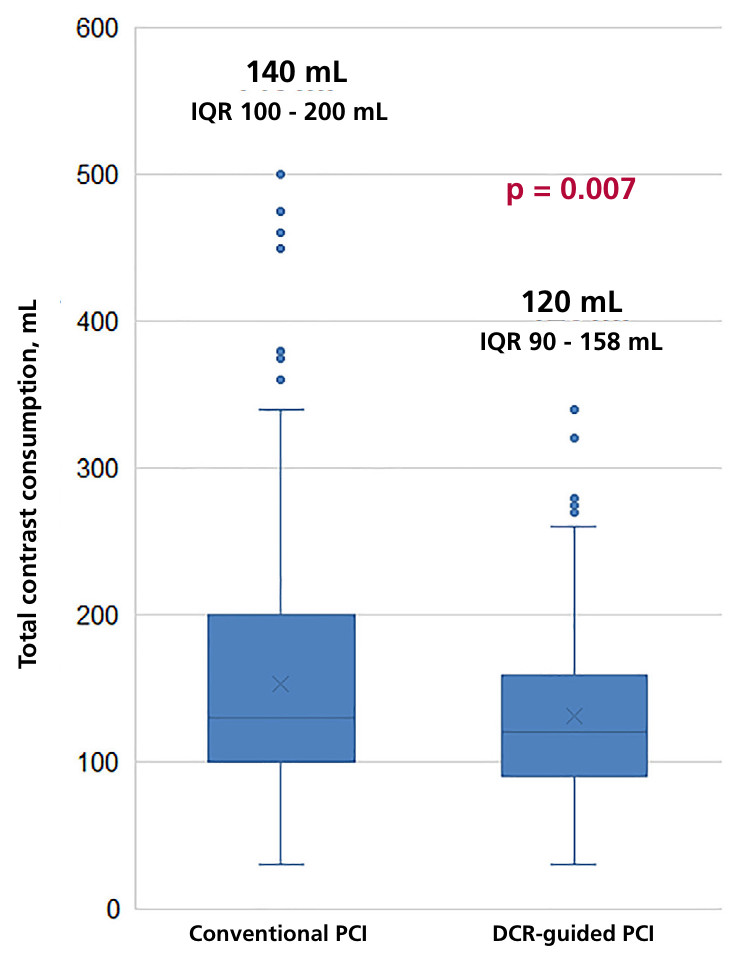
A significant reduction in contrast use was observed in the DCR group (median of 120 mL vs. 140 mL, p = 0.007) (Figure 1). As detailed in Table 3, this difference remains even when stratifying the data according to procedure type and PCI complexity. These findings, adjusted for sex, age, and procedure complexity using a linear regression model, confirm an average reduction of 37.3 mL (95% CI 24.3 -50.5 mL; p <0.0001) of contrast per patient in the DCR group.
Secondary outcomes
Radiation exposure during each procedure was analyzed, expressed in terms of Kerma (an indicator of the amount of radiation emitted and received in the working field, measured in Gy) and Dose-area product (DAP; a dosimetric quantity that expresses the total amount of radiation emitted to the patient and is usually measured in Gy·cm²), with no significant differences observed between the two groups (Table 3). With regard to renal function, given its direct relationship with the use of contrast medium, the variation in serum creatinine (ΔCr) was evaluated, comparing the values prior to the procedure with those recorded before discharge. No significant differences were found between the groups.
Table 3
Radiation, contrast dose, and renal function evolution according to the use or non-use of DCR
| DCR group (n= 201) | Control group (n= 279) | p | |
|---|---|---|---|
| Total contrast, mL | 120 (90-158) | 140 (100-200) | 0.007 |
| Contrast in PCI, mL | 100 (70-140) | 120 (80-180) | 0.001 |
| DAP, Gy.cm 2 | 41.7 (25.3-68) | 45.5 (27.7-69.7) | 0.846 |
| Kerma, mGy | 517 (299-824) | 514 (315-801) | 0.567 |
| Cr delta, mg/dL | 0.10 (0-0.02) | 0.10 (0.01-0.2) | 0.813 |
Cr: creatinine; DAP: dose area product; DCR: Dynamic Coronary Roadmap; PCI: percutaneous coronary intervention.
Variables are expressed as median (interquartile range).
DISCUSSION
To our knowledge, this is the first study in our region to evaluate the impact of using the DCR on the reduction of contrast volume in PCI procedures. Our findings provide relevant evidence on the applicability of this technology in an unselected population with significant comorbidities and complex coronary anatomies.
The main observations were: a) a significant reduction in contrast use in the DCR group (median of 120 mL vs. 140 mL; p=0.007); b) adjusting for age, sex, type of procedure, and PCI complexity, an average reduction of 37.3 mL per patient was observed; c) no significant differences were observed in post-procedural renal function or radiation exposure.
Performing complex procedures in patients with comorbidities and challenging anatomies involves risks that must be minimized. Among these, CIN is a major concern, with an incidence of ≤1% in patients without risk factors, (1,2) but which can rise to 10- 30% in patients with diabetes and/or chronic kidney disease after angiography. (3,4) In addition, recent reviews and international guidelines confirm the relevance of this complication and emphasize the need for preventive strategies. (16-18) In this context, any tool that reduces contrast exposure without compromising the effectiveness of the procedure becomes clinically relevant.
The finding of a reduction in contrast requirements with the use of DCR is consistent with other reports in the literature. (5-8) The multicenter randomized trial DCR4Contrast demonstrated significant reductions in contrast volume and number of angiographic acquisitions. (9,10) Similarly, two recent meta-analyses confirmed that the use of DCR is associated with lower contrast volume and fluoroscopy time, without compromising the success rate of the procedure. (11,12)
Recent studies on low-contrast PTCA techniques highlight the integration of DCR as part of broader strategies to minimize nephrotoxicity. (13,14) Furthermore, recent reviews and international guidelines confirm the relevance of NIC and emphasize the need for preventive strategies. (15-18)
Although no significant differences in serum creatinine variation were observed in our cohort, the reduction in contrast supports the potential of DCR to mitigate adverse effects. A meta-analysis of seven studies (2020-2024) reported a lower incidence of CIN in patients undergoing DCR-guided PCI (OR 0.50; 95% CI 0.27-0.93). (11) Likewise, complementary technologies, such as contrast modulation systems, have been shown to reduce contrast use and the incidence of AKI. (19)
In resource-limited settings, the implementation of DCR may represent an initial challenge in terms of technological investment. However, its potential to reduce contrast consumption, improve procedural efficiency, and decrease renal complications could offset these costs in the mid-term. The progressive availability of consoles integrated into modern angiography systems could favor its regional adoption, especially in centers with a high volume of procedures.
Our study has limitations: being observational, the risk of bias persists despite multivariable adjustment, limiting a definitive causal relationship; in addition, the sample size may not be sufficient to detect differences in rare outcomes such as CIN.
Despite these limitations, our results provide useful evidence in a regional context, where population characteristics and access to advanced technologies can condition the implementation of innovations. In addition to its clinical implications, reducing contrast volume has potential economic benefits, as CIN is associated with higher hospital costs. (20) Resource optimization and improved patient safety are aligned with public health policies aimed at reducing catheterization-related morbidity and improving the efficiency of cardiovascular care.
It is worth mentioning that the adoption of DCR in our center occurred progressively during the first quarter of 2024. No significant differences in contrast volumes were observed over time, suggesting a rapid learning curve for the team and stable integration of the tool into daily practice.
CONCLUSIONS
The use of DCR during PCI was associated with a significant reduction in contrast volume without affecting renal function or increasing radiation exposure. These findings, consistent with previous literature, provide relevant local evidence in a context where socioeconomic variability and access to advanced technologies can influence clinical practice. The reduction in contrast consumption not only has implications for patient safety, but also for the optimization of healthcare costs and resources, aligning with public health strategies aimed at improving efficiency and reducing the morbidity associated with catheterization.
Conflicts of interest
None declared.
(See authors' conflict of interests forms on the web).
Funding
None.