INTRODUCTION
The left atrium (LA) is a structure of fundamental importance for cardiac physiology and hemodynamics. However, for many years it was considered solely as a cavity with the passive function of conducting blood flow from the pulmonary veins to the left ventricle.
There are multiple diagnostic methods for detecting anatomical and functional alterations of the LA, but their usefulness in routine cardiological practice has not been widely disseminated, nor has their implication in the development, progression, and complications of multiple diseases, among which heart failure (HF), atrial fibrillation (AF), and stroke stand out.
To write this review, a thorough search and extensive literature review was conducted on the definition, pathophysiology, diagnosis, and current diagnostic implications of atrial myopathy in patients with heart failure with preserved ejection fraction.
The following sources of medical information were used: bibliographic databases (MEDLINE), evidence-based medicine databases (Cochrane Library, Uptodate), specialty journals, clinical practice guidelines (European Society of Cardiology, American College of Cardiology/American Heart Association), and the central library of the Italian Hospital of Buenos Aires.
The following words and terms were used for the search: "Atrial myopathy," "Atrial cardiomyopathy," "HFpEF," "Heart Failure with preserved ejection fraction," and their equivalents in Spanish.
Definition
The term atrial myopathy first appeared in 1972 following a study published by Nagle et al. (1) in which they described a familial syndrome that predominantly affected the atrium, associated with atrioventricular conduction disorders. An approximation of its definition emerged in 2018, when the European Heart Rhythm Association, together with the Heart Rhythm Society, the Asia Pacific Heart Rhythm Society, and the Latin American Society of Cardiac Rhythm and Electrophysiology, developed a consensus defining atrial myopathy as "any complex of structural, architectural, contractile, or electrophysiological changes affecting the atria that might produce clinically relevant manifestations." (2)
Pathophysiology
The LA performs five primary functions. First, it functions as a cavity that receives blood from the pulmonary veins. Second, it has a contractile function that helps fill the left ventricle at end diastole. Third, it acts as a conduit that admits passive filling of the left ventricle due to pressure differences. Fourth, it has the ability to detect changes in blood volume through baroreceptors, which allows it to regulate the release of natriuretic peptides and thus regulate diuresis. Finally, it has mechanoreceptors that increase the rate of discharges to the sinus node, facilitating tachycardia when venous return increases. (3,4)
Regarding the pathophysiology of atrial myopathy, multiple factors are taken into consideration that can lead to structural and functional deterioration of the LA. Shen et al. (5) propose a staging model of atrial myopathy progression that begins with a normal atrium, on which the effect of factors such as age, inflammation, atrial enlargement and oxidative stress, among others, lead to stage A, represented by a high risk of developing atrial myopathy. These risk factors progress, producing fibrosis, structural remodeling, and electrophysiological alterations (stage B), characterized by asymptomatic atrial myopathy that is detectable by imaging studies. Stage C presents clinical manifestations of reversible atrial myopathy and in stage D, irreversible atrial alterations take place.
Etiology
Multiple etiologies have been associated with the development of atrial dysfunction. The most frequent are: (6)
-
Rhythm or conduction disturbances: they lead to structural and electrical alterations in the atrium. These can be arrhythmias such as AF, or conduction disorders that cause poor atrioventricular electrical coupling, such as complete left bundle branch block or advanced interatrial block.
-
Atrial cardiomyopathy: This corresponds to a group of entities characterized by atrial fibrosis. Among the most frequent are idiopathic atrial fibrosis, atrial ischemia, and atrial involvement secondary to myocarditis.
-
Atrial remodeling: These are conditions that cause structural, electrophysiological, and cellular alterations in the atrial myocardial tissue secondary to an increase in left atrial pressure or volume. Among the most common causes are AF and alterations of the left ventricle or the atrioventricular valves.
Diagnosis
There are currently no specific diagnostic criteria for atrial myopathy. Electrocardiogram (ECG), echocardiogram, and magnetic resonance imaging are among the studies that contribute to the diagnostic approach. It is considered that a histological study of the atrial myocardium is necessary to make an accurate diagnosis of atrial myopathy.(7)
In terms of electrocardiographic changes that point to the identification of atrial myopathy, interatrial block stands out, which can be partial, when the duration of the P wave is equal to or greater than 120 ms, or advanced, when P wave prolongation is accompanied by morphological changes in the inferior leads (biphasic P waves in DII, DIII, and aVF). There are also other changes that have prognostic implications in atrial myopathy, such as the terminal force of the P wave in V1 (>40 000 µV/ms) and the MVP (Morphology-Voltage-P) score. (8)
The MVP score can be obtained through an ECG and is based on three variables: the morphology of the P wave in inferior leads (the morphology of partial interatrial block is equivalent to 1 point and high-grade block to 2 points), the voltage in D1 (between 0.20 and 0.10 mV equals one point, and less than 0.10 mV to 2 points) and the duration of the P wave (between 120 and 140 ms equals one point and greater than 140 ms equals 2 points). The score was initially designed to identify patients at high risk of developing AF: 0 to 2 points represents low risk, 3 to 4 points intermediate risk, and 5 to 6 points high risk. However, the variables it analyzes, such as interatrial block and P wave voltage, are considered markers of atrial myopathy. (9)
Atrial fibrillation, as an entity, may be useful for the diagnostic approach to atrial myopathy. The appearance of f waves on the ECG may be an indicator of electrophysiological and structural changes in the atrial myocardium. There is still debate as to whether AF is a marker of atrial myopathy or whether arrhythmia per se as a pathological entity leads to the development of atrial fibrosis.(7)
The echocardiographic parameters commonly used to assess atrial structure include the anteroposterior length of the LA and the left atrial area and volume (total and indexed to body surface area).(10) Techniques such as Doppler and speckle tracking are used to assess left atrial function.(11)
Conventional echocardiography can be used to perform various calculations based on volumetric measurements to estimate left atrial function. The following calculations can be performed: (12–14)
-
Total LA ejection fraction = [(Maximum LA volume - Minimum LA volume) / Maximum LA volume] × 100. This represents the reservoir function.
-
LA expansion index = [ (Maximum LA volume - Minimum LA volume)/Minimum LA volume] × 100. This also represents the reservoir function.
-
Passive LA emptying fraction = [(Maximum LA volume – LA volume before the P wave in the ECG)/Maximum LA volume] × 100. It represents the conduction function.
-
Active LA emptying fraction = [(LA volume before the P wave in the ECG – LA minimum volume)/LA volume before the P wave in the ECG] × 100. This represents the contraction function.
In addition to the anatomical parameters measured by conventional echocardiography, Doppler can be used to obtain indirect data on LA function, such as peak A wave and, using tissue Doppler, the A´ wave; however, these have the disadvantage of being dependent on loading conditions and age. (13,15)
In the review by F. Triposkiadis et al.,(16) four stages of LA dysfunction in patients with HF are proposed according to echocardiographic parameters.
-
Stage 1: there is prolonged left ventricular relaxation, causing LA impaired passive emptying. On echocardiography, it manifests as preserved LA volumes and pressures with an increased Doppler A wave.
-
Stage 2: there is an increase in LA pressure associated with a decrease in ventricular compliance. Doppler shows increased E wave relative to the A wave (pseudonormalization).
-
Stage 3: there is marked LA dilation, and most ventricular filling occurs in early diastole. Doppler shows a more marked increase in the E wave relative to the A wave.
-
Stage 4: it is mainly characterized by an increased risk of developing AF, with the consequent loss of LA pump function. Doppler shows E waves in the absence of A waves.
Three phases of the cardiac cycle can be measured using longitudinal LA strain: (17)
-
Reservoir strain: it represents the isovolumetric contraction of the left ventricle, ejection, and isovolumetric relaxation. It is calculated by subtracting the peak value of the strain curve from the value at end-diastole, and is represented by a positive value.
-
Conduction strain: it represents the time from mitral valve opening to the start of atrial contraction in patients in sinus rhythm (in patients with AF, it is considered until mitral valve closure). It is calculated by subtracting the strain value at the start of atrial contraction from the peak value of the strain curve, and is represented by a negative value.
-
Contraction strain: it represents atrial contraction in patients with sinus rhythm and is calculated by subtracting the strain value at end diastole from the value at the onset of atrial contraction.
Pathan et al.(18) conducted a systematic review and meta-analysis to determine the normal values for LA strain, including 40 studies (2542 patients) with information on reservoir function; 14 studies (805 patients) with information on conduction function; and 18 studies (1005 patients) with information on contractile function. Analyses were carried out to rule out sources of heterogeneity and the following cut-off points were defined for normality: reservoir strain: 39.4% (95% CI 38.0%–40.8%), conduction strain: 23.0% (95% CI 20.7%–25.2%) and contraction strain 17.4% (95% CI 16.0%–19.0%).
An acceptable correlation has been described between atrial strain parameters measured by echocardiography and invasive measurements of intracavitary pressures. In a study by Lundberg et al.(19) in which 164 patients underwent right heart catheterization for suspected HF or dyspnea of unclear cause, it was concluded that measurement of LA global longitudinal strain is a useful and applicable tool for identifying elevated LA pressures and estimating left ventricular filling pressures. It has even been associated with the development of symptoms in patients in early stages of HF, as in the study published by Maffeis et al.(20) which included 185 patients in stages A and B of HF with preserved ejection fraction. The alterations in echocardiographic parameters such as indexed left ventricular (LV) volume, peak longitudinal strain, and contraction strain had a statistically significant association with the presence of symptoms in both stages.
Patients with HF with preserved ejection fraction often present varying degrees of secondary mitral regurgitation as a result of LA or LV dilation. In the study published by Tamargo et al. (21) including 280 patients undergoing invasive cardiac catheterization for exertional dyspnea evaluation, the prevalence of atrial dysfunction, defined as a reservoir strain below 24.5%, was twice higher in patients with mitral regurgitation compared with patients with competent mitral valves.
Magnetic resonance imaging is a tool of fundamental importance for studying atrial anatomy and function. The presence of late gadolinium enhancement at the LA level has been associated with an elevated risk of developing HF and stroke in this group of patients. In a study published by Zhou et al. (22) including 153 dialysis patients (96 with HF with preserved ejection fraction and 52 healthy controls) undergoing LA strain analysis using magnetic resonance imaging, the diagnostic accuracy for identifying patients with HF with preserved ejection fraction was increased over conventional parameters of atrial geometry and echocardiographic indices.
Figure 1 presents an overview of the different diagnostic methods for approaching atrial myopathy.
Fig. 1.
Diagram of diagnostic methods for atrial myopathy
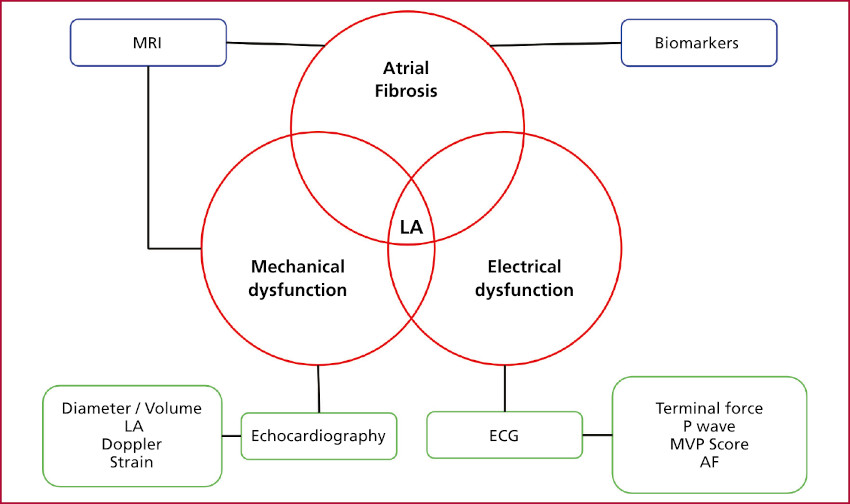
Using a pathophysiological approach to atrial myopathy based on three fundamental pillars—atrial fibrosis, mechanical dysfunction, and electrical dysfunction—an interpretive framework for diagnostic methods is developed.
AF: atrial fibrillation; ECG: electrocardiogram; LA: left atrium; MRI: magnetic resonance imaging; MVP: Morphology-Voltage-P
Prognosis
Structural alterations of the LA are associated with a poor prognosis in patients with HF, regardless of ejection fraction or the presence of arrhythmias. (23,24) In the general population, a 30% increase in left atrial volume is associated with 43% higher risk of developing AF, and in patients over 65 years of age, it doubles the risk of developing HF.(4) In a systematic review and meta-analysis published by Khan et al. (25) that included 2 clinical trials and 20 observational studies with a total population of 1974 symptomatic patients with HF with preserved ejection fraction vs. 751 controls, alterations such as increased LA volume and reduced atrial strain were significantly more marked in patients with HF than in controls. However, this did not translate into greater prognostic capacity for the combined endpoint of all-cause mortality or hospitalizations for HF. On the other hand, in the Heart and Soul Study substudy which included 855 patients with stable coronary artery disease and left ventricular ejection fraction (LVEF) >50%; a decrease in the LA function index was associated with an increased risk of hospitalization for HF, regardless of variables such as age, classic cardiovascular risk factors, and other echocardiographic parameters such as indexed LA volume, LVEF and left ventricular mass index. (26)
Another substudy, published in 2016, considered patients enrolled in the TOPCAT (Spironolactone for Heart Failure with Preserved Ejection Fraction) study. It included 357 patients with HF with preserved ejection fraction and echocardiography image quality sufficient to perform an AI analysis using speckle tracking. Among patients with normal atrial volume, 47% had abnormal atrial strain, compared with 71% of patients with atrial dilation. In an average follow-up of 31 months, lower values of peak atrial strain were associated with the primary composite endpoint of cardiovascular death, hospitalizations for HF, and resuscitated sudden cardiac death, in addition to hospitalizations for HF.(27)
In an analysis of the PARAMOUNT (LCZ696 Compared to Valsartan in Patients With Chronic Heart Failure and Preserved Left-ventricular Ejection Fraction) study, which included symptomatic HF patients with preserved ejection fraction in functional class II – IV, 135 patients with sinus rhythm and echocardiographic images of sufficient quality to perform speckle tracking analysis were compared with 40 controls. Reservoir, conduction, and contraction strain values were significantly lower in patients with HF than in controls, even after adjusting for variables such as indexed LA volume, left ventricular end-diastolic volume, E´ value, and E/A and E/e´ ratios. In addition, lower values of systolic atrial strain were associated with a higher prevalence of hospitalizations for HF and a history of AF.(28)
Freed et al.(10) prospectively enrolled 308 patients who were hospitalized for HF with preserved ejection fraction and underwent echocardiography with speckle tracking analysis. All LA strain parameters were predictors of hospitalization due to cardiovascular disease and death. Reservoir strain maintained prognostic utility regardless of AF history, LA volume, and left ventricular mass.
A systematic review and meta-analysis was recently published, including 25 cohort studies with a total of 252 504 patients, analyzing electrocardiographic, echocardiographic, and laboratory parameters associated with atrial myopathy and stroke. Results show that terminal P wave force in V1, LA diameter, reservoir strain, and N-terminal pro-B-type natriuretic peptide (NT-proBNP) level were statistically associated with an increased risk of ischemic stroke. (29)
Therapeutic implications
There are currently no therapies with proven efficacy for the treatment of atrial myopathy. However, there are therapies for the treatment of closely related conditions such as HF and AF. In patients with symptomatic HF, both the European Society of Cardiology and the American Heart recommend the use of loop diuretics for symptomatic relief. (30,31) In patients with overt volume overload, LA pressure is typically elevated, so diuretic treatment is effective for symptomatic relief. However, the use of diuretics in euvolemic patients with the aim of lowering LA pressures can be harmful and may even lead to worsening renal function. (32)
In 2018, the REDUCE LAP-HF I (Transcatheter Interatrial Shunt Device for the Treatment of Heart Failure with Preserved Ejection Fraction) study was presented, including 94 patients with symptomatic HF with preserved ejection fraction and elevated end-expiratory wedge pressure during exercise testing. Patients were randomly assigned in 1:1 ratio to an interatrial shunt device (Corvia Medical) or control. At one month of follow-up, patients in the treatment arm had a greater reduction in wedge pressure compared with patients in the control group, though not reaching statistical significance, (change in wedge pressure of -3.5±6.4 mm Hg vs. -0.5 ±5.0 mm Hg, p=0.14). This did not translate into significant differences in terms of cardiovascular or cerebrovascular events.(33)
Later in 2022, the REDUCE LAP-HF II (Atrial Shunt Device for Heart Failure with Preserved and Mildly Reduced Ejection Fraction) study was published, in which 626 patients were randomly assigned in 1:1 ratio to placement of an atrial communication device or a placebo procedure. After a median follow-up of 2 years, no significant differences were observed in the composite primary endpoint of cardiovascular death, non-fatal ischemic stroke at 12 months post-randomization, HF events, or change in quality of life as measured by the Kansas City Questionnaire, nor in its individual components. The only point at which a benefit was demonstrated was in the change of functional class, but the patients who received the device experienced adverse cardiac events (cardiac death, myocardial infarction, cardiac tamponade, or emergency cardiac surgery) more frequently than the control group.(34)
CONCLUSIONS
Atrial myopathy is an underdiagnosed clinical entity that has important implications for the diagnosis and prognosis of patients with HF with preserved ejection fraction. However, its usefulness as a specific target for pharmacological and non-pharmacological treatment has not yet been demonstrated.
Funding
None.
Conflicts of interest
None declared.
(See authors' conflict of interests forms on the web).
Acknowledgments
We are deeply grateful to the Institute of Cardiovascular Medicine at Hospital Italiano de Buenos Aires.