Leonardo Caceres 1 MTSAC, Rodrigo Cano 1, Camila Correa Sadouet 1, Adrian Mahl 1, Gisela Streintenberger 1, Guillermo Mazo 1, Maribel Kanchi 1, Heraldo D’Imperio 1
1 Área de Investigación. Sociedad Argentina de Cardiología
Address for reprints: Leonardo Caceres. Área de Investigación de la Sociedad Argentina de Cardiología. E-mail: leo_1208_caceres@gmail.com
Rev Argent Cardiol 2024;92:144-150. http://dx.doi.org/10.7775/rac.v92.i2.20751
ABSTRACT
Background: Hypertrophic cardiomyopathy (HCM) is the most common genetic disease caused by cardiac sarcomere protein mutations, with considerable prevalence and different clinical presentation, varying from asymptomatic to heart failure and sudden death. Some patients are followed-up in nonspecialized centers, and it is necessary to know data that show the reality of their diagnosis, treatment, and prognosis.
Objective: The aim of this study was to know the clinical characteristics, and diagnostic and therapeutic strategies when HCM is managed in centers not specialized in this disease.
Methods: This was a national, cross-sectional, multicenter study, with quantitative analysis of patients with confirmed or highly probable HCM.
Results: A total of 95 patients were recruited, mostly men, with hypertension (40%) and dyslipidemia (22%) as main risk factors. A low proportion of comorbidities was observed: chronic obstructive pulmonary disease (6%), prior myocardial infarction (5%), prior stroke (1%) and chronic kidney failure (1%). The main symptoms were dyspnea (47% and angina (27%), and the most used diagnostic methods were echocardiogram (97%) and cardiac magnetic resonance imaging (71%)). The most frequent localization was septal, with 37% of hypertrophic obstructive cardiomyopathy.
The genetic test, performed in 33% of patients, was positive in more than half of cases. It was not performed in the rest of the patients, mainly due to lack of health coverage.
Conclusions: These findings are in agreement with international registries. Based on our findings, emphasis should be placed in improving the access to more complex diagnostic studies and optimizing the resources in a fragmented health system.
Key words: Hypertrophic cardiomyopathy - Registry - Nonspecialized centers - Clinical practice
RESUMEN
Introducción: La miocardiopatía hipertrófica (MCH), es la enfermedad genética cardiovascular más común, causada por mutaciones en proteínas del sarcómero cardíaco, con una prevalencia considerable y clínica variable, desde asintomática hasta insuficiencia cardíaca y muerte súbita. Existen pacientes seguidos en centros no especializados, y es necesario conocer datos que puedan mostrar la realidad de su diagnóstico, tratamiento y pronóstico.
Objetivo: Conocer las características clínicas, estrategias diagnósticas y terapéuticas al abordar la MCH en centros no especializados en la patología.
Material y métodos: Estudio de corte transversal, multicéntrico, de alcance nacional, con análisis cuantitativo, de pacientes con MCH confirmada o altamente probable.
Resultados: Se registraron 95 pacientes, mayormente hombres con hipertensión arterial (40 %) y dislipidemia (22 %) como principales factores de riesgo. Se observó baja proporción de comorbilidades: enfermedad pulmonar obstructiva crónica (6 %), infarto de miocardio previo (5 %), accidente cerebro vascular previo (1 %) e insuficiencia renal crónica (1 %). Los síntomas principales fueron la disnea (47 %) y el ángor (27 %), y los métodos diagnósticos más usados fueron el ecocardiograma (97 %) y la resonancia cardíaca (71 %). La localización más frecuente fue septal, con 37 % de tipo obstructivo.
El test genético, realizado en un 33 %, fue positivo en más de la mitad de los pacientes. No se realizó en dos tercios de los casos principalmente por falta de cobertura.
Conclusiones: Los hallazgos son concordantes con los de registros internacionales. Con base a nuestros hallazgos, se resalta la necesidad de mejorar el acceso a estudios diagnósticos más complejos y optimizar recursos en un sistema de salud fragmentado.
Palabras clave: Miocardiopatía hipertrófica - Registro - Centros no especializados - Práctica clínica
Recibido: 12/20/2023
Aceptado: 02/05/2024
INTRODUCTION
Hypertrophic cardiomyopathy (HCM) is the most common genetic disease affecting the heart, with an approximate prevalence of 1 in 200/500. (1,2) Its etiology lies in the mutation of a series of genes that codify proteins of the cardiac sarcomere, developing left ventricular hypertrophy (LVH), myofibrillar disarray and myocardial fibrosis. (3,4) Clinical manifestations are variable, from absence of symptoms to heart failure and sudden death. (5,6)
This entity is a veritable diagnostic challenge, as there are infiltrative cardiomyopathies that behave as phenocopies. (5,7)
Currently, pharmacological treatments, implantable devices and surgeries are recommended, improving survival and quality of life. (8-10)
An important number of patients with suspected HCM is followed -up in centers not specialized for the disease. We consider that the data derived from these patients can contribute to reflect in a more comprehensive way the reality of this disease.
We present the first report of the ongoing Hypertrophic Cardiomyopathy Registry in nonspecialized centers in Argentina.
OBJECTIVES
The aim of the study was to know the clinical characteristics of patients, and the diagnostic and therapeutic strategies applied by clinical cardiologists in nonspecialized centers to conditions highly compatible with HCM.
METHODS
This was a national, cross-sectional, multicenter study in patients with confirmed or highly probable HCM, with the participation of clinical cardiologists attending outpatient clinics of nonspecialized centers.
Patients ≥18 years old with confirmed or highly probable diagnosis of HCM established by imaging or laboratory studies (Doppler echocardiography, late gadolinium-enhancement magnetic resonance imaging and/or genetic test), as considered by the clinical cardiologists following them up, were included in the study. Patients with diseases or situations that generated secondary ventricular hypertrophy: hypertension (HTN), valvular diseases, infiltrative cardiomyopathies, athletes, etc, and patients with HCM followed-up in specialized centers, were excluded from the study. A specialized center was defined as a center specialized only in cardiology and/or cardiomyopathies specific outpatient clinics.
Data collection was performed through the REDCap platform of the Argentine Society of Cardiology from June 1 to September 30, 2023.
Statistical analysis
Qualitative variables are presented as frequencies and percentages and quantitative variables are expressed as mean ± standard deviation (SD) or median and interquartile range (IQR 25%-75%), according to their distribution. The analysis of discrete variables was performed using the chi-square test of Fisher´s exact test, as appropriate, and continuous variables were analyzed with the t test or the Mann Whitney test, according to their distribution. A two-tail p<0.05 was considered significative. The R statistical software package was used to perform the analyses.
Ethical considerations
The protocol was approved by the Research Committee of Autonomous City of Buenos Aires Government. An informed consent was waived as it was a study with anonymized data, without personal information.
RESULTS
A total of 95 patients distributed in 8 provinces were included in the study. Mean age was 50 years, with predominance of male sex (58%). The prevalence of cardiovascular risk factors was: HTN 40%, dyslipidemia 22%, obesity 15% and diabetes 14%. A low proportion of comorbidities was observed: chronic obstructive pulmonary disease in 6% of cases, prior myocardial infarction in 5%, prior stroke in 1% and chronic kidney failure in 1%. (Table 1)
A third of patients consulted for the first time with presumptive or confirmed diagnosis of HCM, 37% for some symptom and 27% for a routine control. As shown in Figure 1, dyspnea and angina were the most frequent symptoms, followed by palpitations and syncope. Among patients with dyspnea, 41% were in functional class III/IV, representing 9.5% of all patients.
Regarding diagnostic studies, 96% of patients had an electrocardiogram (90% with sinus rhythm, 91% with signs of left ventricular hypertrophy), 96% an echocardiogram, 71% cardiac magnetic resonance (CMR), and 66% 24-hour ECG Holter monitoring. A genetic test was performed in 33% of patients, coronary angiography in 22%, exercise stress-echo in 13% and coronary computed tomography angiography in 7%. (Figure 2)
Both in the echocardiogram and CMR, LVH was most frequently observed in the interventricular septum (67% and 51%, respectively), and in the apex (9% and 13%, respectively). A significant difference was observed in left ventricular ejection fraction (LVEF) quantification by echocardiography compared with CMR, with median (IQR 25%-75%) of 61% (55%-67%) vs. 67% (65%-74%), respectively, p<0.001. An obstructive gradient (>30 mmHg) of the left ventricular outflow tract (LVOT) was detected at rest in 35 patients (37%) with a mean of 43±26 mmHg at rest and 62±39 mmHg with Valsalva maneuver (p<0.001).
The genetic test was positive in approximately half of the cases in which it was performed (52%). At the time of data collection for the registry, the result was pending in 1 out of 3 patients. The most prevalent genetic alterations identified were TNNT2 (5 patients), MYH7 (5 patients) and MYBPC3 (4 patients). Interrogation of the reasons for not requesting the genetic test revealed that in 19 cases the healthcare plan did not cover the test, in 16 cases the center did not have access to the test, in 12 cases the physicians never request it, in 4 cases the test was refused by the patient and in 12 cases for other causes.
Family members were studied in 44% of cases, by means of echocardiogram (41 cases), CMR (15 cases) and genetic test (14 cases).
Medication consisted mostly of betablockers (91%), calcium channel blockers (41%), angiotensin converting enzyme system inhibitors/angiotensin II receptor antagonists (ACEI/ARAII, 47%) and statins (37%). Figure 3 describes the medication the patients were receiving prior to consultation and that added during follow-up.
Table 1. Baseline data
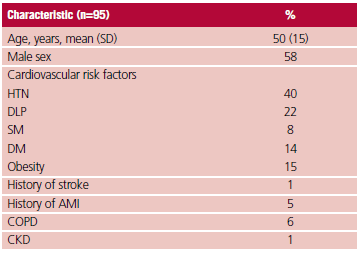
AMI: acute myocardial infarction; DM: diabetes mellitus; DLP: dyslipidemia; COPD: chronic obstructive pulmonary disease; CKD: chronic kidney disease; HTN: hypertension; SD: standard deviation; SM: smoking.
Regarding invasive treatments, among the 22 patients who consulted with confirmed HCM diagnosis or for a second opinion, 45% had some previous invasive treatment: 23% implantable cardioverter defibrillator (ICD), 18% myectomy, 4% pacemaker and 4% alcohol ablation. During follow-up 22 patients required new invasive treatments: ICD 18%, pacemaker 3%, resynchronization therapy 1% and septal myectomy 4%.
DISCUSSION
The present study represents the first approach for the care of patients with HCM in nonspecialized centers of Argentina. Although most cardiologists who follow-up these patients are concentrated in Buenos Aires and Autonomous City of Buenos Aires, there is representation in other provinces: Santa Fe, Formosa, Catamarca, Tucumán, Río Negro, Mendoza and Chubut (see Appendix).
Great progress has been achieved in the field of HCM, both in imaging diagnosis as treatments, providing patients a life expectancy similar to that of the general population. (11) Knowledge of the data of the real world in this rare disease is important to identify the impact of these new strategies. An outstanding example is the Hypertrophic Cardiomyopathy Registry (HCMR), the largest prospective and multicenter study, with 2755 patients from 44 centers in 6 countries, which in addition to clinical data, included CMR characteristics, genotypification and biomarkers. (12- 14) In our setting there is no history of registry of these patients with focus on nonspecialized centers. Among the most important findings of our registry, we noticed that patients were diagnosed at an age similar to that observed in the HCMR, with a similar proportion of hypertensive cases, though with a greater share of diabetics. (12)
Dyspnea was the predominant symptom, and more than 25% of patients were diagnosed after a routine consultation, strongly indicating the high level of suspicion that should be adopted for the diagnosis of this pathology, as many present asymptomatic or oligosymptomatic.
One of the most important contributions of the present registry refers to requested studies. The most frequent are ECG and echocardiogram, in probable relationship to their ample availability in the territory. ECG Holter monitoring was used in 66% of cases, similar to the HCMR (60%). However, exercise echo-stress was scarcely requested, in approximately 1 out of 10 patients, contrary to the suggestion of the 2023 ESC guideline on the management of cardiomyopathies, which recommends it as a IB indication at the time of evaluating the LVOT gradient. (5,15,16)
If we analyze the echocardiographic data, we can observe that the percentage of patients with LVOT obstruction was higher in our registry compared to the HCMR (37% vs. 18%). However, the mean gradient at rest was lower in our study (43±26 mmHg vs. 69±31 mmHg). These trends require an additional validation through the inclusion of a larger number of patients. In the case of CMR, it should be pointed out that a not negligible percentage of patients with highly probable HCM diagnosis lack this study (3 out of 10). This is another data that contradicts the HCMR, where 9 out 10 patients have undergone the study. (12,17)
Nevertheless, the CMR findings are similar, the septal and apical locations being the most frequent and mean LVEF showing similar values (67% vs. 64%). (12,13) An interesting aspect is the variability of LVEF and cardiac mass quantification by echocardiography and CMR, probably related with the spatial resolution of the latter. The genetic test has not performed in 2 out of 3 patients, and the most common reason for not requesting it by the follow-up physician is lack of resources or health coverage. (18)
Regarding pharmacological treatment, use of betablockers and calcium channel blockers was greater in our registry compared with the HCMR (91% vs. 57% and 27% vs. 18.7%, respectively), which could be due to the difference in patients with LVOT obstruction and in functional class III/IV (37% vs. 18% and 9.5% vs. 7.2%). The same trend is observed in the use of ACEI/ARAII and statins (47% vs. 23.7% and 37% vs. 27%). It should be pointed out that 48% of patients did not present with pharmacological treatment prior to consultation with the follow-up physician. (12)
Invasive treatments in HCM are part of the mainstays in the management of these patients, especially in those who continue to be symptomatic despite the pharmacological treatment or are at risk of sudden death. (5,19,20). In our registry, 1 out of 10 admitted patients, already had some treatment prior to the first consultation with the treating physician, the most important being ICD. One-fourth of patients required intensive care during follow-up, with predominance of implantable devices. The low rate of myectomy and alcohol ablation should be pointed out, despite the percentage of obstructive type and functional class III/IV patients.
The specially fragmented healthcare system in Argentina, in addition to the inequity of resources available in different provinces of our territory, might explain the differences with other countries. This point could help us optimize the resources in this type of disease.
As limitations, the sample size of this registry is not representative of all the Argentine territory. Moreover, it has weaknesses inherent to retrospective registries with voluntary participation, which might generate reporting biases.
Fig. 1. Consultation symptoms (n=95).
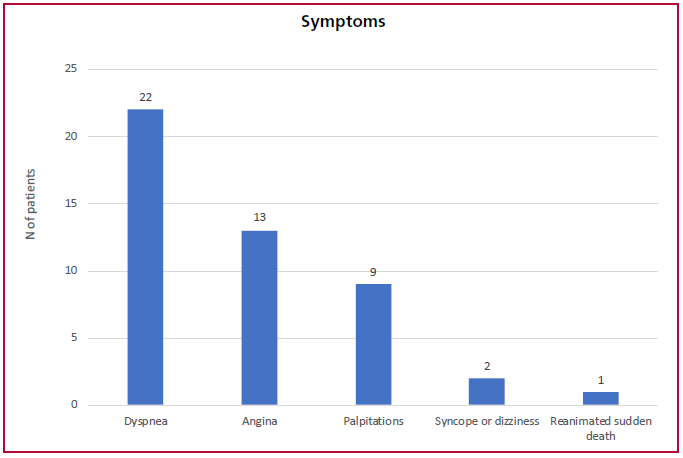
Fig. 2. Studies performed.
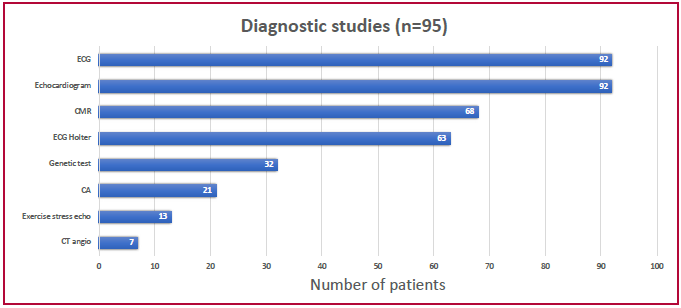
CA: Coronary angiography; CMR: Cardiac magnetic resonance; CT angio: CT angiography; ECG: Electrocardiogram.
Fig. 3. Prior and added medication
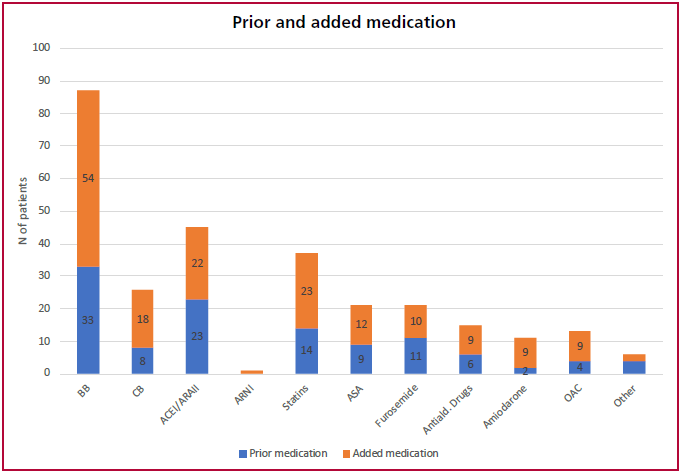
ACEI: angiotensin-converting enzyme inhibitors; ARA II: angiotensin II receptor antagonists Antiald: antialdosterone drugs; ARNI: angiotensin receptor-neprilysin inhibitor; ASA: acetylsalicylic acid; BB: betablockers; CB: calcium channel blockers; OAC: Oral anticoagulation.
CONCLUSIONS
The present registry is the first study of patients with HCM in Argentina which provides data of the real world in the care of patients in nonspecialized centers. Dyspnea and chest pain were the most frequent symptoms of clinical presentation. The ECG and echocardiogram stand out as the pillars for diagnostic suspicion, and in this sense, there is a space for improvement for the access to more complex studies. Pharmacological treatment is in agreement with that established by international guidelines.
Conflicts of interest
None declared.
(See conflicts of interest forms on the website).
https://creativecommons.org/licenses/by-nc-sa/4.0/

©Revista Argentina de Cardiología
BIBLIOGRAFÍA
- Khorana AA, Francis CW, Culakova E, Kuderer NM, Lyman GH. Thromboembolism is a leading cause of death in cancer patients receiving outpatient chemotherapy. J Thromb Haemost. 2007;5:632-4. https://doi.org/10.1111/j.1538-7836.2007.02374.x
- Zou Y, Song L, Wang Z, Ma A, Liu T, Gu H, et al. Prevalence of idiopathic hypertrophic cardiomyopathy in China: a population-based echocardiographic analysis of 8080 adults. Am J Med. 2004;116:14-8. https://doi.org./10.1016/j.amjmed.2003.05.009
- Maron BJ, Desai MY, Nishimura RA, Spirito P, Rakowski H, Towbin JA, et al. Diagnosis and Evaluation of Hypertrophic Cardiomyopathy: JACC State-of-the-Art Review. J Am Coll Cardiol. 2022;79:372-89. https://doi.org/10.1016/j.jacc.2021.12.002
- Zipes DP, Libby P, Bonow RO, Mann DL, & Tomaselli GF. Braunwald. Tratado de cardiología: Texto de medicina cardiovascular. Elsevier Health Sciences (2019).
- Baxi AJ, Restrepo CS, Vargas D, Marmol-Velez A, Ocazionez D, Murillo H. Hypertrophic Cardiomyopathy from A to Z: Genetics, Pathophysiology, Imaging, and Management. RadioGraphics 2016;36:335–54. https://doi.org/10.1148/rg.2016150137
- Arbelo E, Protonotarios A, Gimeno JR, Arbustini E, Barriales- Villa R, Basso C, et al; ESC Scientific Document Group. 2023 ESC Guidelines for the management of cardiomyopathies. Eur Heart J. 2023;44:3503-626. https://doi.org/10.1093/eurheartj/ehad194
- Maron BJ, Rowin EJ, Udelson JE, Maron MS. Clinical Spectrum and Management of Heart Failure in Hypertrophic Cardiomyopathy. JACC: Heart Failure 2018;6:353–63. https://doi.org/10.1016/j.jchf.2017.09.011
- Rowin EJ, Maron BJ, Maron MS. The Hypertrophic Cardiomyopathy Phenotype Viewed Through the Prism of Multimodality Imaging: Clinical and Etiologic Implications. JACC Cardiovasc Imaging. 2020;13:2002-16. https://doi.org/10.1016/j.jcmg.2019.09.020
- Fernández A, Acunzo RS, Avegliano G, Casabé JH, Dumont CA, Hita A, Ortiz M, Pérez de Arenaza D, y cols. Consenso Argentino de diagnóstico y tratamiento de la Miocardiopatía Hipertrófica 2016. Sociedad Argentina de Cardiología. Rev Argent Cardiol 2017;85(Suplemento 2):1-78.
- Kotkar KD, Said SM, Dearani JA, Schaff HVl. “Hypertrophic obstructive cardiomyopathy: the Mayo Clinic experience.” Annals of Cardiothoracic Surgery 2017:6:329-36. https://doi.org/10.21037%2Facs.2017.07.03
- Hodges K, Rivas CG, Aguilera J, Borden R, Alashi A, Blackstone EH, et al. “Surgical management of left ventricular outflow tract obstruction in a specialized hypertrophic obstructive cardiomyopathy center.” The Journal of Thoracic and Cardiovascular Surgery 2019:157:2289–99. https://doi.org/10.1016/j.jtcvs.2018.11.148
- Maron, B. J., Rowin, E. J., Casey, S. A., & Maron, M. S. How hypertrophic cardiomyopathy became a contemporary treatable genetic disease with low mortality: shaped by 50 years of clinical research and practice. JAMA cardiology 2016;1:98-105. https://doi.org/10.1001/jamacardio.2015.0354
- Neubauer S, Kolm P, Ho CY, Kwong RY, Desai MY, Dolman Sf, et al; HCMR Investigators. Distinct Subgroups in Hypertrophic Cardiomyopathy in the NHLBI HCM Registry. J Am Coll Cardiol. 2019;74:2333-45. https://doi.org/10.1016/j.jacc.2019.08.1057
- Wigle ED, Sasson Z, Henderson MA, Ruddy TD, Fulop J, Rakowski H, et al. Hypertrophic cardiomyopathy. The importance of the site and the extent of hypertrophy. A review. Progress in Cardiovascular Diseases .1985;28(1):1-83. https://doi.org/10.1016/0033-0620(85)90024-6
- Kramer CM, Appelbaum E, Desai MY, Desvigne-Nickens P, DiMarco JP, Friedrich MG, et al. Hypertrophic Cardiomyopathy Registry: The rationale and design of an international, observational study of hypertrophic cardiomyopathy. Am Heart J. 2015;170:223-30. https://doi.org/10.1016/j.ahj.2015.05.013
- Ommen SR, Mital S, Burke MA, Day SM, Deswal A, Elliott P, et al. 2020 AHA/ACC Guideline for the Diagnosis and Treatment of Patients With Hypertrophic Cardiomyopathy: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2020;76:159-240. https://doi.org/10.1016/j.jacc.2020.08.045
- Shah JS, Esteban MT, Thaman R, Sharma R, Mist B, Pantazis A, et al. Prevalence of exercise-induced left ventricular outflow tract obstruction in symptomatic patients with non-obstructive hypertrophic cardiomyopathy. Heart 2008;94:1288–1294. https://doi.org/10.1136/hrt.2007.126003
- Maron MS, Olivotto I, Zenovich AG, Link MS, Pandian NG, Kuvin JT, et al. Hypertrophic cardiomyopathy is predominantly a disease of left ventricular outflow tract obstruction. Circulation CIRCULATIONAHA.106.644682
- Rudolph A, Abdel-Aty H, Bohl S, Boye P, Zagrosek A, Dietz R, et al. Noninvasive detection of fibrosis applying contrast-enhanced cardiac magnetic resonance in different forms of left ventricular hypertrophy relation to remodeling. J Am Coll Cardiol 2009;53: 284-291. https://doi.org/10.1016/j.jacc.2008.08.064
- Richard P, Charron P, Carrier L, Ledeuil C, Cheav T, Pichereau C, et al. Hypertrophic cardiomyopathy: distribution of disease genes, spectrum of mutations, and implications for a molecular diagnosis strategy. Circulation 2003;107:2227–2232. https://doi.org/10.1161/01.CIR.0000066323.15244.5419. Maron BJ, Spirito P, Shen WK, Haas TS, Formisano F, Link MS, et al. Implantable cardioverter-defibrillators and prevention of sudden cardiac death in hypertrophic cardiomyopathy. JAMA 2007;298:405–412. https://doi.org/10.1001/jama.298.4.405
- Robbins RC, Stinson EB. Long-term results of left ventricular myotomy and myectomy for obstructive hypertrophic cardiomyopathy. J Thorac Cardiovasc Surg 1996;111: 586-594. https://doi.org/10.1016/S0022-5223(96)70310-0
