INTRODUCTION
Carotid artery atherosclerosis is the most common cause of extracranial carotid artery disease and is one of the leading causes of cerebrovascular disease (CVD) and death, (1,2) constituting the predominant etiology of CVD in Western countries. (3) However, other non-atheromatous causes of carotid disease are also related with CVD such as fibrodysplasia, Takayasu disease, and aortic dissection. (1,4) Dolichoarteriopathies (from the Greek δόλιχος, dolichos, "abnormally long") have recently attracted the attention of specialists and are included within non-atheromatous carotid artery diseases. Carotid dolichoarteriopathies (CDA) have been associated with a wide range of CVD, including stroke and transient ischemic attack (TIA). (1) However, their clinical significance remains unclear. (1) This article reviews the main aspects of CDA, focusing on its pathophysiology, diagnosis, prognosis, and treatment.
METHODS
We conducted a non-systematic review and selected the most relevant papers on this topic. PubMed, Scielo, and Scholar Google electronic databases were searched using the terms "dolichoartheriopathies," "carotid dolichoartheriopathies," "arterial tortuosity," "dolichoectasia," and their equivalents in Spanish.
RESULTS
Definition, classification and epidemiology
CDA are anatomical anomalies that result in a change in the geometric arrangement of the carotid vessels along their course, that causes coiling, kinking, and tortuosity. (1,5) Some authors include them within the group of dolichoectasias (vessel elongation, tortuosity and/or dilatation). (6) First described by Kelly in 1925, (7) CDA were classified by Weibel and Fields, (8) in three groups (Figure 1):
-
Type I or tortuosity: any S-shaped or C-shaped elongation or undulation of the course of the artery with an angulation >90°.
-
Type II or coiling: the internal carotid artery forms an exaggerated, redundant S-shaped curve or its longitudinal axis makes a complete circle (360°).
-
Type III or kinking: Angulation of one or more segments of the artery, with an angle < 90°.
-
Metz et al. sub-classified kinking based on the value of the acute angle between the two segments forming the kink in group I (between 90° and 60°, Figure 2, Panel A), group II (between 60° and 30°, Figure 2, Panel B) and group III (>30°, Figure 2, Panel C). (9)
Figure 1.
Classification of carotid dolichoarteriopathies diagram combining the Weibel-Fields and Metz classifications. The Weibel-Fields classification diagram has been modified from Reference #41. The Metz classification illustration has been modified from Reference #5
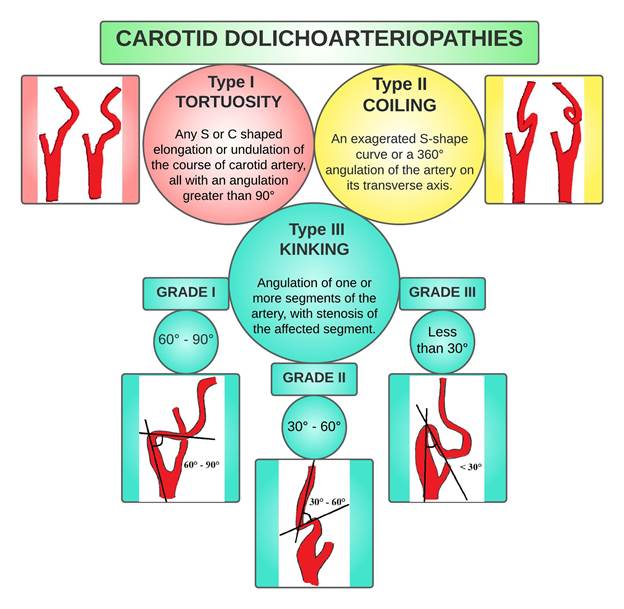
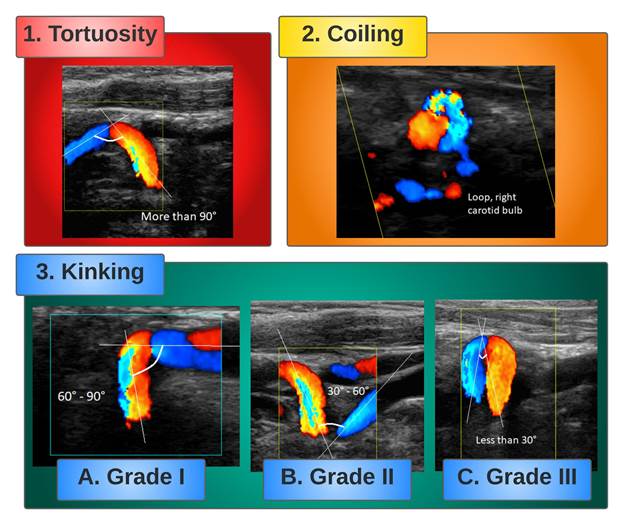
Figure 2.
Images of different types of carotid dolichoarteriopathies obtained with color Doppler ultrasound. The angle between each part of the artery is drawn with white lines and described in the corresponding image.
CDA are common, with a reported prevalence of 10-45%. (10) Beigelman et al. (1), and Ghilardi et al. (11) made the diagnosis of CDA in 13.3-31% of the cases, while in studies conducted in hospitalized patients, the prevalence ranged between 13.5% and 58%. (6) They are more prevalent in women and elderly patients, especially those > 60 years, and can be unilateral or bilateral, affecting the common carotid artery (CCA), internal carotid artery (ICA) and external carotid artery (ECA), but more commonly the ICA, especially on the left side. (12,13) Tortuosity and kinking are more common than coiling. (1,12)
Pathophysiology
The development of CDA has been linked to several mechanisms, although there is still some debate about the exact causes. A key question is whether they are congenital or acquired and whether they are related to atherosclerosis or other cardiovascular risk factors. Some studies shed light on the subject.
A. Embryology
The cerebral vasculature begins to develop early in the embryo, during the third week of intrauterine life, with the formation of the aortic arches (AA). (13,14) There are six pairs of AA connecting the ventral aorta (VA) to the dorsal aorta (DA). (14,15) Each AA arises from the bulbus arteriosus, and courses in its corresponding pharyngeal arch to end in the DA. (14) The first and second arches regress, and their remnants from the VA and DA persist as incipient ICA and ECA. The ICA merges bilaterally with the third AA, while the ventral aortic root between the third and fourth arches persists as the CCA. (15) There is considerable variability in the course and position of the CCA and carotid bifurcations. Elongation of the CCA and ICA results in tortuosity and kinking. (16) The position of the carotid bifurcation reflects the degree of embryologic migration of the ECA and is variable. (16) Huber et al. reported the position of the carotid bifurcation at C4 to C5 in 48% and at C3 to C4 in 34% of 658 bifurcations. (17) There are cases described of bifurcations from T3 to C2. (18) In children, bifurcation position from C2 to C3 was observed in 40% of cases and from C3 to C4 in 40% of cases. (19) Carney et al. found tortuosity in 5 of 20 fetuses of ages ranging from 5 months to full term. (20) Harrison et al. suggested that the development of carotid arteries and skeletal system might be asynchronous, causing changes and tortuosity in the course of the artery. (21) Beigelman et al. studied a population of 885 individuals ranging from newborns (4 hours 30 minutes) to 90 years. The study population was divided into two groups: a control group of 245 individuals (children and adolescents up to 15 years old) and another group of 640 individuals (16-90 years old) in whom diagnostic color Doppler ultrasonography of the neck vessels had been requested because of clinical suspicion of atherosclerotic vascular disease. The incidence of coiling and kinking was similar in both groups. The presence of atheromatous plaques in tortuous arteries was only observed in 3 group 2 patients. The authors found a lack of correlation with other cardiovascular risk factors, and of the site of carotid artery plaques with CDA. (1)
B. Genetic and molecular disorders
Voevoda et al. examined 61 families of children with previously diagnosed ICA dolichoarteriopathy using color Duplex ultrasound. The group comprised 100 individuals with ICA dolichoarteriopathy. Control group (n=245) was formed from DNA bank based on a population approach. The authors found an association between A80807T polymorphism of the transcriptional factor Sp4 gene and CDA. (22) Zaidi et al. reported the case of a consanguineous Kurdish family whose child had severe elongation and tortuosity of the aorta, carotid and other arteries, along with other changes (loose skin, joint hypermobility, hernias, and facial features resembling Ehler-Danlos syndrome [EDS]). By homozygosity mapping, they found evidence of the involvement of the locus at chromosome 20q13 in the affected child. (23) Arslan et al. observed significantly higher matrix metalloproteinase-2 (MMP-2) levels in patients with CDA compared to the control group. The expression of MMP-12 was higher in those with atheromatous plaques than in those without atherosclerosis. (24)
C. Connective tissue diseases (CTD)
Foiadelli et al. reported the case of 7 children with EDS between 3 and 13 years of age, with CDA and variable clinical manifestations. (25) Welby et al. evaluated 286 patients divided into control group and CTD group. The presence of CDA was 44% in the second group, and 16% in controls (p < 0.001). Coiling was more prevalent. The prevalence of CDA was 88% in Marfan syndrome, 63% in Loeys-Dietz syndrome, 42% in neurofibromatosis type 1, and 19% in patients with both vascular and nonvascular types of EDS. (26)
D. Association with other congenital diseases
According to Paltseva et al., CDA have higher elastin levels but with fiber fragmentation, higher MMP-9 expression, and lower smooth muscle actin expression. (27) Ballotta et al. analyzed 78 carotid arteries with dolichoarteriopathy and found typical and atypical patterns of fibromuscular dysplasia (FMD). (28) Sethi et al. found a significantly higher prevalence of CDA with S-shaped curve in patients with FMD. (29)
Ballota et al. analyzed 43 patients with asymptomatic abdominal aortic aneurysm (AAA) and CDA and found degenerative dysplastic changes in the tunica media in all carotid specimens; in a few cases, nonobstructive atherosclerotic intimal lesions were superimposed. The histologic features of classic AAA (thinning of the media underlying the atherosclerotic plaque) were observed in almost all aortic wall specimens, suggesting underlying connective tissue disorders in both conditions. (30)
E. Acquired mechanisms
Harrison et al. suggest that CDAs may be due to kyphosis or lordosis, which would deviate the carotid axis. (21) For Etheredge et al., inflammation of the pericarotid tissues would cause retraction of the carotid arteries and deviate them from their path, resulting in tortuosity. (31)
Ghilardi et al. and Del Corso et al. describe a high prevalence of hypertension (HTN) and atherosclerosis in patients with CDA. (11,32) However, both studies lacked a control group of normal subjects and addressed only a population selected because of vascular disease, in which these cardiovascular risk factors are expected to predominate. For Khasiyev et al., because CDA also show dilatation, compensatory external remodeling in response to atherosclerosis may play a role in this phenotype. (6) Dilatation of the carotid arteries may also be a sign of connective tissue weakness of a non-atherosclerotic nature. Pancera et al. found a statistically significant association between kinking and HTN, and also between HTN and kinking with a greater degree of bending. (33) Oliviero et al. analyzed the prevalence of CDA in patients with HTN and diabetes. The prevalence of CDA was significantly higher in the group of hypertensive patients than in diabetics and controls and was also associated with the duration of hypertension. (34) The eccentric forces related to HTN and possibly artery aging with stiffness may play a role in ICA dilatation and, thus, in the development of CDA. (6)
Wang et al. evaluated the association between body mass index (BMI) and CDA. They analyzed 513 patients without CTD using the tortuosity index (TI) expressed as a percentage, calculated as the ratio between the actual length of the ICA (AL) and the length measured from the carotid bifurcation to the base of the skull, called the cord length (CL):
Male sex and BMI were significantly correlated with the TI. For each increase in BMI of 1 kg/m2, there was a 1.59-fold increase in the risk of developing CDA. (35) Dilba et al. used the same index in a population of the Plaque At RISK study and concluded that age, obesity and hypercholesterolemia were associated with higher TI. They also found higher prevalence and severity of CDA in the left ICA. Higher BMI and CDA could be due to high intra-abdominal pressure which may result in an upward shift of the diaphragm and a rise of the mediastinum. This would push up the carotid arteries, which, surrounded by lax connective tissue, can slip and become tortuous. Because the brachiocephalic artery "absorbs" part of the lifting force, the right ICA develops less severe tortuosity than the left. Thus, repeated and prolonged exposure to periods of high and low flow results in stress that affects arterial remodeling and causes tortuosity. (36)
Derubertis et al. observed a higher prevalence of CDA in previously irradiated patients. Radiation can cause retraction and alteration of the connective tissue, resulting in tortuosity of the artery. (37) Saba et al. analyzed a population of 124 patients with ICA dissection diagnosed by computed tomography (CT) or magnetic resonance imaging (MRI) and found a statistically significant association between dissection and kinking and coiling. (38) Barbour et al. found a significant association between spontaneous ICA dissection and tortuosity, particularly if tortuosity is present bilaterally. (39) Some authors propose that changes in hemodynamics might lead to the local disruption of the endothelial integrity which trigger inflammation that can modify the structure of the vessel, and the release of inflammatory agents might predispose to carotid dissection. (6)
Diagnosis
A diagnosis of CDA can be made by many imaging tests. Ultrasound seems to be the gold standard. (1,2,6) The turbulent blood flow inside the tortuous vessels can be evaluated with Doppler scanning. (40) Di Pino et al. studied the prevalence of dolichoarteriopathies of the ICA using color Doppler ultrasound in 2856 subjects (0-96 years) and demonstrated peaks of prevalence distributed in a bimodal fashion (< 21 years and > 60 years). CDA were detected in 9.9% of participants. This study was one of the first to use ultrasound in a large cohort. (41) Uchino et al. reported a case of CDA diagnosed by computed tomography (CT) angiography and single photon emission computed tomography (SPECT). Both tests better defined the morphology of ICA tortuosity and secondary perfusion abnormalities. (42) In other studies, CT allowed the evaluation of subtle abnormalities, such as microaneurysms, and 3D reconstructions of CT appear to be more effective in classifying morphological variations of the ICA and detecting these anomalies. (43-45) Balevi et al. found a prevalence of CDA of 40.3% in the general population using contrast-enhanced magnetic resonance angiography (MRA). (12) Tomiya et al. performed MRA in 13 patients and obtained very clear images of CDA. (46) Yu et al. believe that MRA is a good option for the diagnosis of CDAs. Digital subtraction angiography (DSA) remains the gold standard for the diagnosis of cervical and intracranial vascular disease, could provide hemodynamic data and show the morphology of CDAs in detail, but cannot determine the pathological changes of the arterial wall of the ICA. (40) It would serve as a complementary imaging test in the study of CDAs rather than a useful imaging test for diagnosis. All these techniques are valid and not mutually exclusive and should be chosen in order of increasing complexity and invasiveness to make a correct diagnosis.
Clinical implication
The clinical implication of CDA is controversial. There is limited evidence to support the association between CDA and CVD. (6) Yin et al. observed a higher incidence of white matter lesions in patients with CDA compared to controls. The severity of these lesions was directly related to the number of kinks. (47) Other authors argue that ischemic disease is more common in people with CDA and other cardiovascular risk factors. Pancera et al. found a significant association between kinking and TIA. Hypertension and CDA could be additive risk factors in the pathophysiology of TIA. (33) Oliviero et al. demonstrated that the presence of carotid kinking in hypertensive subjects may not be considered a further risk factor for ischemic events. (48) But, as Pancera et al. state, the addition of risk factors to the presence of CDA may increase the risk of vascular events. (33) Iwai-Takano et al. observed an association between CDA and aging, HTN and sex, but not with dyslipidemias, diabetes or smoking. (49)
Those who consider that CDA are harmful believe that kinking in the ICA, in addition to causing luminal narrowing and endothelial lesions, can create turbulent blood flow that can lead to a prothrombotic state. For others, as Balevi et al., CDA are more a curiosity than a real predictor of ischemic events. (12) Valvano et al. believe that there is no association between CDA and CVD. (50) CDA can cause cerebral hypoperfusion that produces encephalopathy, vertigo, diplopia, TIA or infarction. (40) The prevalence of cerebrovascular symptoms in patients with CDA varies between 15 and 23%. (51) Coiling is not considered a risk factor for ischemic events due to its weak association with symptoms, as opposed to kinking which might be associated with symptoms which most commonly appear due to transitory hypotension during sleep or sudden and extreme movement of the head and neck. (40) Other symptoms observed are visual impairment as amaurosis fugax, uveitis, retinitis and macular dystrophy. (52) Severe cases of CDA can produce mass effect, resulting in dysphagia, dyspnea, upper airway obstruction and obstructive apnea, and, more rarely, pulsatile tinnitus and hemilingual spasm. (40,53,54)
In the Northern Manhattan Study (NOMAS), Khasiyev et al. analyzed the relationship between CDA and biomarkers of atherosclerosis in 558 participants and found an association between CDA and diastolic blood pressure and larger aortic root diameter, but not with other markers of atherosclerosis. Determining the risks of vascular events associated with this non-atherosclerotic phenotype may help for a better risk stratification for individuals with CDA. (55)
Beigelman et al. investigated the genesis of neurological complications associated with CDA. Sixty patients with CDA without atherosclerosis underwent head rotation tests and color Doppler ultrasound of the ICAs and ophthalmic arteries with evaluation of flow velocities. The results suggested that CDA do not cause neurological abnormalities or symptoms, as no events were recorded during the test, and a significant reduction in the velocities in the ophthalmic artery was observed in only 3 of 60 cases. (56)
Wang et al. analyzed the blood pressure changes caused by CDA in 12 patients using DSA. They measured the angle of kinking by rotational angiography and calculated the blood pressure at the proximal, kinking and distal sites of the ICA using a microcatheter. These results were compared with two simulation models. A numerical simulation model was constructed with a software based on a geometric model of a tortuous ICA. An in vitro fluid simulation model was built using a silicone tube with different kinking angles. Water flowed through the silicon tube via a peristaltic pump. The software simulation showed a linear drop in blood flow pressure with decreases in the kinking angle. The pressure drop was the smallest at the kinking angle of 180° and was the largest at 30°. However, a reversal occurred between the kinking angles of 30° and 20°; the increasing kinking degree led to a smaller pressure drop. This was confirmed in the in vitro experimental system. When the flow rate was constant, the pressure drop decreased with increasing kinking angle, but the pressure drop increased when the kinking angle increased from 20° to 30°. However, these findings did not correlate with clinical observations: the clinical tests performed on patients showed greater pressure drops when the kinking angle was < 30°, and severe kinking induced an average decrease of 15.5% in blood pressure. The development of the reversal of greater pressure drops with increased kinking angles from 20° to 30° was not observed in the clinical measurements. This was attributed to excessive elongation and stenosis of the ICA in cases of severe kinking. And although the pressure drop changed gradually in the simulation models, it decreased rapidly in clinical measurements when the angle was < 45º but was more gradual with higher angles. This behavior could be caused by the characteristics of the medium (blood is a non-Newtonian fluid), and the elastic walls of the ICA. Both factors can contribute to the self-regulatory mechanism of blood flow. (57) This was one of the first studies to evaluate these aspects of CDA by comparing clinical and artificial observations with direct measurements of ICA pressures. These results show that blood pressure and blood flow abnormalities may be responsible for the interruption of cerebral perfusion, resulting in ischemia and thus cerebrovascular events.
Some authors observed an association between CDA and aneurysms in the context of CTD. (26) The positive predictive value of the combination of aortic aneurysm and CDA associated with CTD was 95.4% with a specificity of 98.6%. Increased tortuosity index in the aorta and vertebral artery is an independent predictor of aortic dissection in patients with Marfan syndrome. Thus, in patients with CTD, CDA can indicate the presence of clinically significant aneurysms. (6,26,40)
Dolichoarteriopathies have been associated with increased morbidity and mortality in head and neck surgery. This risk was recognized in the studies by Weibel and Fields (8) and Metz. (9) Nayak et al. emphasize the importance of evaluating the morphology of the carotid arteries prior to pharyngeal procedures, as they can be complicated by vascular injury, bleeding, and in severe cases, death due to massive hemorrhage. (58) For this reason, it is essential to thoroughly examine the carotid arteries before performing any procedure in the neck.
Treatment
Despite the controversy about the clinical implications of CDAs, some indications have been developed to manage them according to the degree of hemodynamic involvement of the tortuosity, among other parameters. Gavrilenko et al. (59) proposed treating CDAs under the following conditions:
-
ICA stenosis ≥ 60% with atherosclerotic plaques and any degree of cerebrovascular insufficiency; or
-
ICA stenosis < 60% with atherosclerotic plaques, a moderate to severe degree of cerebrovascular insufficiency in combination with tortuosity, a linear blood flow rate ≥ 110 cm/s and a turbulent blood flow.
Grego et al. (60) proposed specific indications for surgery:
-
TIA (hemispheric symptoms);
-
Asymptomatic patients with kink angle < 30°, with contralateral carotid artery occlusion;
-
Patients with non-hemispheric symptoms after a screening to exclude all other possible neurological or non-neurological causes with: 1) Doppler ultrasound positive for significant increase of flow velocity, 2) positive cerebral CT scan or MRA for ischemic lesions in the ipsilateral hemisphere, and 3) reversal of circulatory flow in the anterior cerebral artery and its reduction in the middle cerebral artery, in relation to rotation and cephalic flexion-extension maneuvers.
Other authors have suggested initiating treatment in cases with an ICA-to-common carotid ratio > 2 or a blood flow velocity greater than 180 cm/s. (61,62) However, the actual indication for treating CDA remains controversial. Most authors agree that symptomatic CDA should be treated after excluding other causes.
Invasive techniques have been proposed to correct CDA with successful results (fixation of the ICA to the digastric muscle, end-to-end anastomosis, end-to-side reimplantation, carotid endarterectomy with patch, bypass grafting, and carotid angioplasty with stenting). (40) However, despite the success of these techniques, the appropriate treatment remains controversial. Further studies are needed to determine the real effectiveness of the surgical approach to CDAs.
CONCLUSION
CDA are a common condition with multifactorial etiology and limited, but not negligible, evidence of cerebrovascular involvement. Further studies are needed to determine the usefulness of the therapeutic strategies to improve its prognosis. Strict monitoring and closer follow-up of patients with global assessment of thromboembolic risk are reasonable measures to prevent future events.
Conflicts of interest
None declared. (See authors conflicts of interest forms in the website).
Financing
None
REFERENCES
1. Beigelman R, Izaguirre AM, Robles M, Grana DR, Ambrosio G, Milei J. Are kinking and coiling of carotid artery congenital or acquired? Angiology. 2010;61:107-12. https://doi.org/10.1177/0003319709336417
2. Beigelman RL, Izaguirre AM, Azzato F, Milei J. Carotid Artery - Pathology, Plaque Structure - Relationship between Histological Assessment, Color Doppler Ultrasonography and Magnetic Resonance Imaging - Dolichoarteriopathies - Barorreceptors. In: Rezzani R, editor. Carotid Artery Disease - From Bench to Bedside and Beyond [Internet]. London: IntechOpen; 2014. https://doi.org/10.5772/57157
3. The global burden of disease: 2004 update. World Health Organization (WHO) Press; 2008. http://www.nhlbi.nih.gov/health/health-topics/topics/catd/[Online]
5. Dilba K, van Dam-Nolen DHK, Crombag GAJC, van der Kolk AG, Koudstaal PJ, Nederkoorn PJ, et al. Dolichoarteriopathies of the extracranial internal carotid artery: The Plaque At RISK study. Eur J Neurol 2021;28:3133-8. https://doi.org/10.1111/ene.14982
6. Khasiyev F, Gutierrez J. Cervical Carotid Artery Dolichoectasia as a Marker of Increased Vascular Risk. J Neuroimaging. 2021;31:251-60. https://doi.org/10.1111/jon.12815
7. Kelly AB. Tortuosity of the internal carotid in relation to the pharynx. J Laring 1925;40:15-20. https://doi.org/10.1177%2F003591572401700607
8. Weibel J, Fields WS. Tortuosity, coiling and kinking of the internal carotid artery.Etiology and radiographic anatomy. Neurology.1965;15:7-18. https://doi.org/10.1212/wnl.15.1.7
9. Metz H, Murray-Leslie RM, Bannister RG, Bull JW, Marshall J. Kinking of the internal carotid artery. Lancet. 1961;1:424-6. https://doi.org/10.1016/s0140-6736(61)90004-6
10. Verlato F, Camporese G, Rocco S, Salmistraro G, Signorini GP. Carotid kinks relationship to atherosclerosis and cerebral ischemia. J Vasc Surg. 2000;32:293-8. https://doi.org/10.1067/mva.2000.106953
11. Ghilardi G, De Monti M, Longhi F, Trimarchi S, Bortolani E. Prevalenza del kinking carotideo in unapopolazioneresidente. Risultatiparzialidelprogramma OPI (ObiettivoPrevenzione Ictus) [Prevalence of carotid kinking in a resident population. Partial results of the OPI (Objective Prevention of Ictus)]. Minerva Cardioangiol. 1993;41:129-32.
12. Balevi M. Dolicho-Arteriopathies (Kinking, Coiling, Tortuosity) of the Carotid Arteries: A Study by Magnetic Resonance Angiography. Int J Neck Head Surg, 2020;11:50-4. https://doi.org/10.5005/jp-journals-10001-1405
13. Valvano A, Bosso G, Apuzzi V, Mercurio V, Di Simone V, Panicara V, et al. Long-term follow-up in high risk hypertensive patients with carotid dolicoarteriopathies. Int Angiol. 2020;39:24-8. https://doi.org/10.23736/S0392-9590.19.04229-9
14. Bertulli L, Robert T. Embryological development of the human cranio-facial arterial system: a pictorial review. Surg Radiol Anat. 2021;43:961-73. https://doi.org/10.1007/s00276-021-02684-y
18. Vitek JJ, Reaves P. Thoracic bifurcation of the common carotid artery. Neuroradiology. 1973;5:133-9. https://doi.org/10.1007/BF00341527
23. Zaidi SH, Peltekova V, Meyer S, Lindinger A, Paterson AD, Tsui LC, et al. A family exhibiting arterial tortuosity syndrome displays homozygosity for markers in the arterial tortuosity locus at chromosome 20q13. Clin Genet 2005;67:183-8. https://doi.org/10.1111/j.1399-0004.2004.00391.x.
24. Arslan Y, Arslan IB, Pekçevik Y, Sener U, Köse S, Zorlu Y. Matrix Metalloproteinase Levels in Cervical and Intracranial Carotid Dolichoarteriopathies. J Stroke Cerebrovasc Dis 2016;25:2153-8. https://doi.org/10.1016/j.jstrokecerebrovasdis.2016.05.028
25. Foiadelli T, Ippolito R, Corbetta R, Simoncelli AM, Amariti R, Licari A, et al. Clinical variability in children with dolichoarteriopathies of the internal carotid artery. Childs Nerv Syst 2020;36:621-8. https://doi.org/10.1007/s00381-019-04395-7.
26. Welby JP, Kim ST, Carr CM, Lehman VT, Rydberg CH, Wald JT, et al. Carotid Artery Tortuosity Is Associated with Connective Tissue Diseases. AJNR Am J Neuroradiol 2019;40:1738-43. https://doi.org/10.3174/ajnr.A6218
27. Paltseva EM, Oskolkova SA, Polyakova VO, Krylova YS, Ivanova AG, Abramyan AV, et al. [The structure of the internal carotid artery wall in pathological tortuosity]. Arkh Patol 2015;77:3-8. Russian. https://doi.og/10.17116/patol20157753-8
28. Ballotta E, Thiene G, Baracchini C, Ermani M, Militello C, Da Giau G, et al. Surgical vs medical treatment for isolated internal carotid artery elongation with coiling or kinking in symptomatic patients: a prospective randomized clinical study. J Vasc Surg 2005;42:838-46; discussion 846. https://doi.org/10.1016/j.jvs.2005.07.034
29. Sethi SS, Lau JF, Godbold J, Gustavson S, Olin JW. The S curve: a novel morphological finding in the internal carotid artery in patients with fibromuscular dysplasia. Vasc Med 2014;19:356-62. https://doi.org/10.1177/1358863X14547122
31. Etheredge SN, Effeney DJ, Ehrenfeld WK. Symptomatic extrinsic compression of the cervical carotid artery. Arch Neurol 1984;41:672-3. https://doi.org/10.1001/archneur.1984.04210080084020
32. Del Corso L, Moruzzo D, Conte B, Agelli M, Romanelli AM, Pastine F, et al. Tortuosity, kinking, and coiling of the carotid artery: expression of atherosclerosis or aging? Angiology 1998;49:361-71. https://doi.org/10.1177/000331979804900505
33. Pancera P, Ribul M, Presciuttini B, Lechi A. Prevalence of carotid artery kinking in 590 consecutive subjects evaluated by Echocolordoppler. Is there a correlation with arterial hypertension? J Intern Med 2000;248:7-12. https://doi.org/10.1046/j.1365-2796.2000.00611.x
34. Oliviero U, Cocozza M, Picano T, Policino S, Russo N, Fazzio S, et al. Prevalence of carotid kinking and coiling in a population at risk.Vasc Surg 1997;31:43-9. https://doi.org/10.1177/153857449703100106
35. Wang HF, Wang DM, Wang JJ, Wang LJ, Lu J, Qi P, et al. Extracranial Internal Carotid Artery Tortuosity and Body Mass Index. Front Neurol. 2017;8:508. https://doi.org/10.3389/fneur.2017.00508
36. Dilba K, van Dam-Nolen DHK, Crombag GAJC, van der Kolk AG, Koudstaal PJ, Nederkoorn PJ, et al. Dolichoarteriopathies of the extracranial internal carotid artery: The Plaque At RISK study. Eur J Neurol 2021;28:3133-8. https://doi.org/10.1111/ene.14982
37. Derubertis BG, Hynecek RL, Kent KC, Faries PL. Carotid tortuosity in patients with prior cervical radiation: increased technical challenge during carotid stenting. Vasc Endovascular Surg. 2011;45:619-26. https://doi.org/10.1177/1538574411408745
38. Saba L, Argiolas GM, Sumer S, Siotto P, Raz E, Sanfilippo R, et al. Association between internal carotid artery dissection and arterial tortuosity. Neuroradiology 2015;57:149-53. https://doi.org/10.1007/s00234-014-1436-x.
39. Barbour PJ, Castaldo JE, Rae-Grant AD, Gee W, Reed JF 3rd, Jenny D, et al. Internal carotid artery redundancy is significantly associated with dissection. Stroke. 1994;25:1201-6. https://doi.org/10.1161/01.str.25.6.1201.
40. Yu J, Qu L, Xu B, Wang S, Li C, Xu X, et al. Current Understanding of Dolichoarteriopathies of the Internal Carotid Artery: A Review. Int J Med Sci. 2017;14:772-84. https://doi.org/10.7150/ijms.19229
41. Di Pino L, Franchina AG, Costa S, Gangi S, Strano F, Ragusa M, et al. Prevalence and morphological changes of carotid kinking and coiling in growth: an echo-color Doppler study of 2856 subjects between aged 0 to 96 years. Int J Cardiovasc Imaging. 2021;37:479-84. https://doi.org/10.1007/s10554-020-02014-0
42. Uchino A, Kohyama S. Long segment dolichoectasia of the right internal carotid artery diagnosed by CT angiography. Radiol Case Rep. 2019;14:1084-7. https://doi.org/10.1016/j.radcr.2019.06.013
43. Mamedov FR, Arutiunov NV, Usachev DIu, Lukshin VA, Beliaev AIu, Mel'nikova-Pitskhelauri TV, et al. [Neuroradiological diagnostics of atherosclerotic lesions and kinking of the carotid arteries in determination of indications to surgical treatment]. Zh Vopr Neirokhir Im N N Burdenko. 2011;75:3-10.
44. Nagata T, Masumoto K, Hayashi Y, Watanabe Y, Kato Y, Katou F. Three-dimensional computed tomographic analysis of variations of the carotid artery. J Craniomaxillofac Surg. 2016;44:734-42. https://doi.org/10.1016/j.jcms.2016.02.011
46. Tomiya Y, Chiba S, Moriyama H, Kikuchi Y, Ohta M. [Eighteen cases of tortuosity of the internal carotid--usefulness of MR-angiography in diagnosis]. Nihon Jibiinkoka Gakkai Kaiho. 1995;98:1367-72. Japanese. https://doi.org/10.3950/jibiinkoka.98.1367
47. Yin L, Li Q, Zhang L, Qian W, Liu X. Correlation between cervical artery kinking and white matter lesions. Clin Neurol Neurosurg. 2017;157:51-4. https://doi.org/10.1016/j.clineuro.2017.04.003
48. Oliviero U, Scherillo G, Casaburi C, Di Martino M, Di Gianni A, Serpico R, et al. Prospective evaluation of hypertensive patients with carotid kinking and coiling: an ultrasonographic 7-year study. Angiology 2003;54:169-75. https://doi.org/10.1177/000331970305400205
49. Iwai-Takano M, Watanabe T, Ohira T. Common carotid artery kinking is a predictor of cardiovascular events: A long-term follow-up study using carotid ultrasonography. Echocardiography 2019;36:2227-33. https://doi.org/10.1111/echo.14536
50. Valvano A, Bosso G, Apuzzi V, Mercurio V, Di Simone V, Panicara V, et al. Long-term follow-up in high risk hypertensive patients with carotid dolicoarteriopathies. Int Angiol. 2020;39:24-8. https://doi.org/10.23736/S0392-9590.19.04229-9
51. Metz H, Murray-Leslie RM, Bannister RG, Bull JW, Marshall J. Kinking of the internal carotid artery. Lancet. 1961;1:424-6. https://doi.org/10.1016/s0140-6736(61)90004-6
53. Sismanis A, Girevendoulis A. Pulsatile tinnitus associated with internal carotid artery morphologic abnormalities. Otol Neurotol. 2008;29:1032-6. https://doi.org/10.1097/MAO.0b013e3181865913
55. Khasiyev F, Rundek T, Di Tullio MR, Wright CB, Sacco RL, Elkind MSV, et al. Systemic Arterial Correlates of Cervical Carotid Artery Tortuosity : The Northern Manhattan Study. Clin Neuroradiol. 2022;32:435-43. https://doi.org/10.1007/s00062-021-01044-y
57. Wang L, Zhao F, Wang D, Hu S, Liu J, Zhou Z, et al. Pressure Drop in Tortuosity/Kinking of the Internal Carotid Artery: Simulation and Clinical Investigation. Biomed Res Int. 2016;2016:2428970. https://doi.org/10.1155/2016/2428970
58. Nayak SB, Shetty SD. Surgical and embryological perspective of a big loop of internal carotid artery extending laterally beyond internal jugular vein. Surg Radiol Anat. 2021;43:413-16. https://doi.org/10.1007/s00276-020-02619-z
59. Gavrilenko AV, Kuklin AV, Khripkov AS, Abramian AV. Otsenka éffektivnosti rekonstruktivnykh operatsii na sonnykh arteriiakh v sochetanii stenoza i patologicheskoi izvitosti [Assessment of efficacy of reconstructive operations on carotid arteries in combination of stenosis and pathological tortuosity]. Angiol Sosud Khir. 2014;20:116-22. Russian.
61. Illuminati G, Ricco JB, Caliò FG, D'Urso A, Ceccanei G, Vietri F. Results in a consecutive series of 83 surgical corrections of symptomatic stenotic kinking of the internal carotid artery. Surgery. 2008;143:134-9. https://doi.org/10.1016/j.surg.2007.07.029
