INTRODUCTION
Heart transplantation is recognized as the optimal therapeutic option for patients with end-stage heart disease.
Transplant rejection and infections are the most frequent complications during the first year post- transplantation. Since rejection may result in graft loss, early detection is crucial to initiate timely and appropriate treatment. 1
In 2021, an observational and retrospective study conducted in our country reported findings consistent with international data, showing that acute rejection was the leading cause of hospitalization during the first year post-transplantation, being cellular rejection the most predominant. 2
Acute rejection encompasses a broad spectrum of presentations, ranging from hemodynamic deterioration to genetic alterations, each with different possible diagnosis. 3 (Figure 1)
Fig. 1.
Diagnostic methods for acute transplant rejection according to the spectrum of presentation.
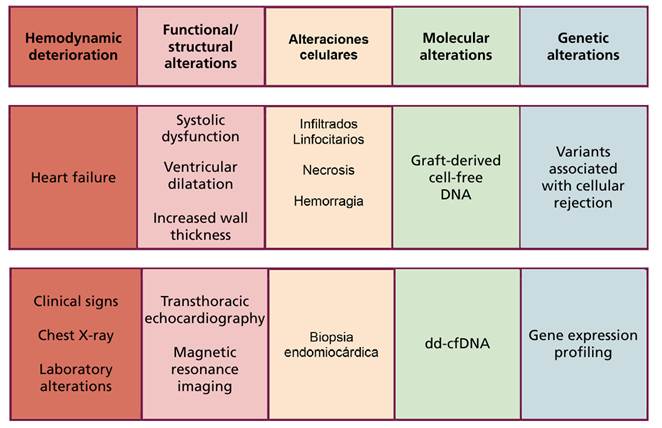
dd-cfDNA, donor-derived cell-free DNA.
Hemodynamic deterioration, most commonly presenting as heart failure, can be diagnosed through a thorough medical history and physical examination, as well as through basic diagnostic tests, such as laboratory analysis and chest X-ray.
Imaging studies can identify structural and/or functional alterations. Echocardiography and magnetic resonance imaging (MRI) can detect increased myocardial thickness, diastolic dysfunction, ventricular dilatation and systolic dysfunction, which may be suggestive of rejection, but, unfortunately, at a more advanced stage than desired, since the objective is to anticipate structural organ damage and initiate early treatment. 4,5,6,7,8,9
For this reason, endomyocardial biopsy (EMB) has become a routine diagnostic procedure. Endomyocardial biopsy reveals microscopically different degrees of cellular infiltrate, necrosis, and/or hemorrhage, which are indicators of rejection. Moreover, it allows for grading the severity of rejection and guiding appropriate treatment.
At the molecular level, the release of graft-derived deoxyribonucleic acid (DNA) can be determined by quantifying the percentage of donor-derived cell-free DNA (dd-cfDNA), representing a diagnostic alternative for identifying rejection.
Finally, at the genetic level, specific variants associated with rejection have been identified. Gene expression profiling (GEP) allows for the identification of patients at higher risk of rejection.
Within the clinical-genetic spectrum, the closer we get to the molecular level, the earlier we can detect the event. 10,11,12
PROBLEM STATEMENT
The most widely used and cost-effective technique for diagnosing and monitoring transplant rejection is the EMB. Although it has adequate sensitivity and specificity for detecting cellular rejection, it has certain limitations, including its invasiveness, potential complications, inconsistency in grading intermediate levels of cellular rejection and challenges in interpreting antibody-mediated rejection.
Among the complications of EMB, cardiac tamponade-although rare (<0.5%)-and the potential to induce tricuspid regurgitation must be mentioned. 13,14
A key factor to consider is that current immunosuppressive therapy has significantly reduced the incidence of acute rejection. Additionally, most routine EMB procedures are performed in asymptomatic patients with a low probability of rejection, which further reduces the diagnostic performance of this technique. 15
Over the last few decades, a reduction in the incidence of rejection has been observed across all intervals post-transplantation. A comparative observational study of two groups of transplant recipients-those transplanted between 1990 and 2000, and those transplanted between 2000 and 2010, found that although the latter group had more risk factors for developing rejection, the incidence was lower in this group at all intervals post-transplantation (0-6 months: 60.2% vs. 21.5%, p<0.001; 6-12 months: 26.8% vs. 1.8%, p<0.001; 12-36 months: 32.3% vs. 10.5%, p=0,006). 16
The decline in the rejection incidence, particularly after the sixth month, markedly reduces the probability of its detection by EMB in asymptomatic patients. It can be concluded that during early post-transplant stages, rejection remains the leading cause of hospitalization and mortality, and that the current diagnostic method is invasive, involves complications, and its diagnostic performance decreases after six months postransplantation in asymptomatic patients, who represent the majority. These factors underscore the need for new strategies to enhance this scenario.
The methods discussed in this article focus on detecting molecular and genetic alterations, beyond the scope of the microscope.
MOLECULAR AND GENETIC DIAGNOSTIC TECHNIQUES
Donor-derived cell-free DNA (dd-cfDNA)
Under normal conditions, the continuous turnover of cells in various tissues results in the presence of circulating cell-free DNA in the bloodstream. The principle underlying the dd-cfDNA technique is the release of donor genetic material into the recipient's blood. Consequently, the detection and quantification of this material may raise alerts during the patient follow-up. It is well established that rejection induces the release of greater amounts of the donor DNA, leading to elevated levels in the recipient's blood. Donor-derived cell-free DNA is expressed as the percentage of the donor DNA relative to the total cell-free DNA in the analyzed sample. The term liquid biopsy has become widely accepted to describe this technique.
A comparative study of dd-cfDNA and EMB showed the normal value of dd-cfDNA and its correlation with the histological grade of rejection. This study also demonstrated that an increase in the dd-cfDNA percentage precedes to histological evidence of rejection. 17
In 2018, the Donor-Derived Cell-Free DNA-Out- comes AlloMap Registry (D-OAR) research group found that dd-cfDNA had a higher negative predictive value (NPV) than EMB after 55 days post-transplantation. 18
In an observational study, the Genomic Research Alliance for Transplantation (GRAfT) included 171 transplanted recipients monitored by EMB and dd-cfDNA. It was found that 80% of patients with dd- cfDNA levels suggestive of rejection (value >0.25%), had not been identified by EMB, and the vast majority of these patients subsequently developed clinically evident rejection during follow-up. 19,20
The DEDUCE study demonstrated that, after the second year of follow-up, dd-cfDNA levels progres- sively increased and were associated with the development of graft vasculopathy, reducing the sensitive of acute rejection diagnosis. 21
Patients with higher grade of cellular rejection exhibit a greater percentage of cell-free DNA. Antibody- mediated rejection is also associated with elevated levels of cell-free DNA as the rejection grade increases. These levels are typically higher than those observed in cellular rejection and are characterized by a distinct amino acid composition.
A consistent finding across all studies is that this technique has a high NPV, approximately 97-99%, and has led to a reduction in the number of EMB procedures. EMBs were performed only when dd-cfDNA levels were markedly elevated, to confirm rejection and categorize its severity.
In conclusion, this technique may be used after the early post-transplant stage, from the second or third month after transplantation. Its high NPV primarily facilitates the exclusion of both cellular and antibody- mediated rejection, even earlier than EMB, and is associated with a decreased frequency of routine EMB procedures. (22-26)
Gene expression profiling (GEP)
The way in which our genes are expressed determines the functional phenotype of all cells, including immune function.
The expression of certain lymphocyte genes has been associated with an increased response and a higher likelihood of cellular rejection.
The most widely used commercial tests detect the presence of 11 genes associated with rejection, establishing a risk score based on their activation profile.
Some of the genes involved include CD3E (lymphocyte activation) and NKG7 (cytotoxicity), among others. 27
A prospective study by the International Society for Heart and Lung Transplantation (ISHLT) showed that more than 55 days post-transplantation, heart transplant recipients with a low gene expression profiling (GEP) score did not experience rejection with hemodynamic compromise, death or retransplantation. This validated the use of GEP in populations at low immune risk.
This technique has demonstrated a high NPV (around 98%), but a low positive predictive value (PPV), although not inferior to that of routine EMB procedure.
As with the dd-cfDNA technique, fewer EMB procedures were performed in patients monitored by GEP. 28
The cut-off point at which the GEP score is considered elevated varies, depending on the baseline immune risk (e.g. sensitized patients) and time post-transplantation. It must be determined serially, since it may vary over time. 29,30,31
Regarding the limitations of GEP, it does not allow determining whether damage is occurring at the time of assessment, nor is it useful for diagnosing antibody- mediated rejection, as it identifies genetic variants in the lymphocytes, which are responsible for cellular rejection. 32,33
Combination of diagnostic tests
According to the evidence analyzed to date on molecular and genetic tests, each technique has been used individually, but they can be used in combination.
The ISHLT published a prospective, observational, multicenter study that included 2077 patients who underwent both tests (GEP and dd-cfDNA) between 2018 and 2021. It is important to note that this study only refers to cellular rejection.
The results were classified as follows:
-
Double negative
-
Positive GEP / negative dd-cfDNA
-
Negative GEP / positive dd-cfDNA
-
Double positive
The incidence of cellular rejection was very low in the double-negative group, 1.5%, compared to 22% in the double-positive group, with intermediate values observed in the other groups.
Using this strategy, the number of EMB procedures decreased significantly (8.8% in the double-negative group) compared to previous levels, and the 2-year survival rate was 94.9%. Only 2.7% of patients experienced graft dysfunction.
The author’s interpretation of the results is shown in Figure 2. If both tests are negative, rejection is con- sidered unlikely, whereas if both tests are positive, rejection is considered highly probable. When GEP is positive and dd-cfDNA is negative, other causes of im- mune activation, such as infections, should be considered. When only dd-cfDNA is positive, cellular or an- tibody-mediated rejection must be ruled out. 31,32
Clinical applicability and areas of uncertainty
Is EMB the gold standard for diagnosing acute heart transplant rejection?
According to the references analyzed, it could be said that EMB is the complementary study with the greatest sensitivity and specificity during the first months post-transplantation. Beyond the disadvantages due to its invasiveness and potential complications, it can be said that after 3 months and even more after 6 months post- transplantation, EMB loses sensitivity, leaving its complications as the main concern.
Another aspect to highlight is that the assessment of the myocardium is partial, as we only examine small samples, and although the inflammation caused by rejection is systemic, it starts in patches. Therefore, in response to the first question, it can be accepted that during the first 3 months post-transplantation, EMB continues to be the gold standard. After this period, GEP and dd-cfDNA techniques have shown excellent NPV for ruling out rejection, thus reducing the need for invasive diagnostic tests.
Regarding the ability to detect rejection, dd-cfDNA technique has shown, in some registries, better sensitivity and specificity than GEP, and also the ability to detect both cellular and antibody-mediated rejection.
Gene expression profiling indicates whether there is activation of the immune system, but it does not provide information about tissue injury. Therefore, it would be a good complement to dd-cfDNA technique. Both techniques have been shown to reduce the number of biopsies per person-year during follow-up.
It is important to note that most studies included patients at low immune risk; they were not recent transplant recipients nor were they sensitized. Consequently, the incidence of rejection in these studies was very low, which means that we do not know the sensitivity and specificity in a general population of transplant recipients.
There are no studies that assess their performance beyond 5 years post-transplantation. However, this is not a clinical concern. From 6 months to 5 years post- transplantation, the analyzed studies showed good results, and rejection can be detected even earlier than with EMB.
The IMAGE study assessed, through a questionnaire, how the different diagnostic tests impacted patients’ quality of life, showing markedly favorable results for the less invasive techniques.
Considering the analyzed information, a surveillance scheme can be proposed combining the different diagnostic techniques, adjusting each one to its best diagnostic profile at different stages of follow-up.
Thus, from the immediate postoperative period up to 2-3 months post-transplantation, EMB has been shown
to be the best method to detect rejection, since all new trials were conducted in patients at least 55 days post-transplantation.
After this period, a scheme using the new surveillance techniques would allow for earlier detection of rejection and a reduction in the number of EMB.
This scheme has been validated only in patients at low immune risk and asymptomatic. In cases of
symptoms or imaging suggestive of rejection, EMB should be performed.
Theoretically, GEP and dd-cfDNA determination could allow titration of immunosuppressants. Evidence is still lacking, but some ongoing studies, such as MOSAIC, are exploring this objective. 34
Both dd-cfDNA and GEP are being used in many transplantation centers, but not yet in Argentina, where economic limitations exist.
In most of these studies, these techniques were compared with EMB as the gold standard. Conducting larger studies with clinical endpoints, such as mortality or retransplantation, is still a pending issue.
Undoubtedly, these techniques provide valuable information, but several aspects remain to be clarified:
-
Cut off point for dd-cfDNA.
-
Cut-off point for GEP.
-
Value at an earlier post-transplant stage.
-
Value in sensitized population.
-
Should a positive dd-cfDNA be treated as rejection?
-
Is it possible to adjust the immunosuppressants dosing based on the values of these techniques?
-
General access to these tests.
-
Availability of study results including clinical end- points.
Considering the available information on heart transplant rejection, it can be said, paraphrasing Antoine de Saint-Exupéry, that “what is essential may be invisible to the microscope.”
Conflicts of interest
None declared. (See authors' conflict of interests forms on the web/Additional material).
