INTRODUCTION
Hypertrophic cardiomyopathy (HCM) is a common and heterogeneous genetic disease, with an estimated prevalence of 1 in 200 to 1 in 500 people. It can present with variable symptoms and serious complications such as heart failure, arrhythmias, and sudden death. (1) It is classified as obstructive HCM (OHCM), characterized by dynamic obstruction of the left ventricular outflow tract (LVOT), and non-obstructive HCM (NOHCM). Although conventional treatments exist, some patients have refractory symptoms or require invasive procedures. In this context, myosin inhibitors have emerged as a new therapeutic strategy to address the underlying pathophysiological mechanisms of the disease. (2,3). This article will review their mechanism of action, the available evidence, indications and forms of use, as well as the associated adverse effects.
MECHANISMS OF ACTION
Myocardial hypercontractility is one of the central characteristics of HCM. This phenomenon results from alterations in actin-myosin interaction, often caused by mutations in sarcomeric genes. In addition to contributing to dynamic obstruction of the LVOT, hypercontractility promotes the development of myofibrillar disorders and myocardial fibrosis. (4,5)
Myosin antagonists, such as mavacamten and aficamten, act by directly modulating actin-myosin interaction, reducing the force of contraction and attenuating the chronic hypercontractile state of myocytes. These drugs selectively inhibit cardiac myosin ATPase, decreasing the formation of cross-bridges between actin and myosin, which results in lower myocardial contractility. In addition, they stabilize the super-relaxed state of myosin, reduce the release of inorganic phosphate, and limit the interaction of myosin with actin, counteracting the hypercontractility observed in HCM. (6) (Figure 1)
Fig. 1
Mecanism of action of aficamten and mavacamten /ADP: adenosine diphosphate; HCM: Hypertrophic cardiomyopathy; MI: myosin inhibitors; Pi: inorganic phosphate
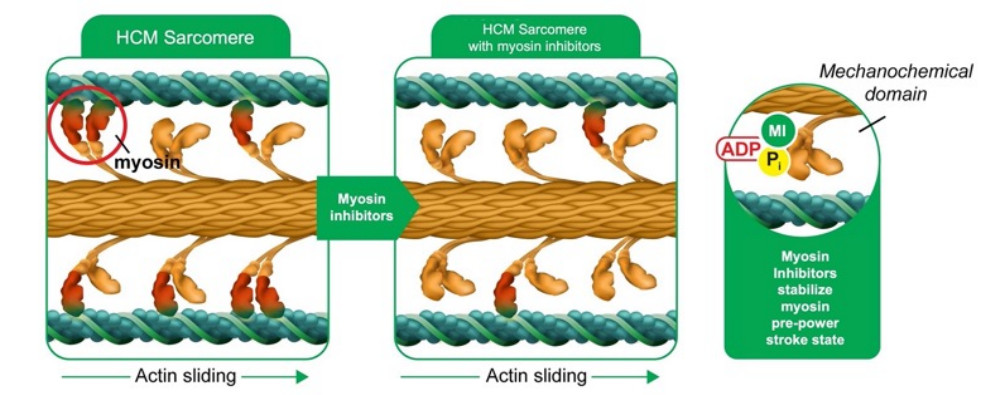
These agents increase the proportion of myosin molecules in a super-relaxed state and promote the kinetics of cross-bridge detachment, stimulate the release of adenosine diphosphate (ADP) from myosin, and promote the binding of adenosine triphosphate (ATP) to myosin. As a result, they not only decrease contractile force but also improve cardiac muscle relaxation, simultaneously addressing hypercontractility and diastolic dysfunction, two of the main pathophysiological mechanisms of HCM. (7-9)
PHARMACOLOGY
Mavacamten is a selective and reversible inhibitor of cardiac myosin ATPase, developed to modulate myocardial contractility. It is administered orally, in doses of 2.5 to 15 mg, with adequate bioavailability and a half-life of approximately 9 days, allowing once-daily dosing. Its metabolism is predominantly hepatic, mediated by CYP2C19 and, to a lesser extent, by CYP3A4 and CYP2C9. This makes it susceptible to interactions with inhibitors and inducers of these enzymes, which can affect its plasma concentration and efficacy. (10)
Aficamten is a second-generation inhibitor of cardiac myosin ATPase. Its dose is 5 to 20 mg, and its half-life is shorter than that of mavacamten (28 to 40 hours), allowing for faster dosage adjustments and a lower risk of accumulation. It is also administered orally and is metabolized in the liver, mainly by CYP3A4, with a lesser contribution from other cytochrome P450 enzymes. Compared with mavacamten, it has fewer relevant drug interactions and a more predictable safety profile, with no need for dose adjustment based on CYP2C19. (10,11)
Available studies suggest that neither drug is significantly excreted renally. However, the pharmacokinetics in patients with severe kidney impairment is not fully characterized. In the presence of moderate to severe hepatic impairment, drug accumulation may occur, although clinical data in these patients are limited.
Mavacamten and aficamten are contraindicated during pregnancy due to their mechanism of action, which may affect fetal cardiovascular development. Therefore, effective contraceptive methods are recommended for women of childbearing age during treatment and for at least four months after discontinuation.
EVIDENCE IN PATIENTS WITH OHCM
Clinical trials
The clinical development of mavacamten began with the phase 2 PIONEER-HCM study, which demonstrated a significant reduction in LVOT obstruction gradient, both at rest and after exercise in patients with symptomatic OHCM. Patients treated with mavacamten experienced improvement in NYHA functional class and a reduction in N-terminal pro-B-type natriuretic peptide (NT-proBNP). In addition, peak oxygen consumption (peak VO2) increased by an average of 1.5 mL/kg/min, reflecting an enhanced functional capacity. (12)
These results led to the phase 3 EXPLORER-HCM trial, which confirmed the efficacy of mavacamten in reducing LVOT gradient, with an average decrease of 40 mmHg.
In addition, 65% of treated patients showed improvement in at least one NYHA functional class, and peak VO2 increased by 1.4 mL/kg/min compared with placebo. (13) In echocardiographic terms, 80.9% of patients in the mavacamten arm achieved complete resolution of systolic anterior mitral valve motion at 30 weeks, suggesting a significant impact on the pathophysiology of the disease. (14)
The phase 3 VALOR-HCM study evaluated mavacamten in patients with severe OHCM with an indication for septal reduction therapy. After 16 weeks of treatment, 82% of patients in the mavacamten arm no longer met criteria for intervention, compared with only 23% in the placebo group. A reduction in LVOT gradient from 51 mmHg to 14 mmHg was observed, with improvement in NYHA functional class and an increase in peak VO2 of 1.7 mL/kg/min. (15)
Long-term data emerge from the MAVA-LTE study, which included patients previously treated in the EXPLORER-HCM trial, with 80-week follow-up. The results confirmed a progressive and sustained reduction in the LVOT gradients, from 48 mmHg to 10 mmHg, as well as a significant decrease in NT-proBNP from 783 ng/L to 123 ng/L. In addition, 60.2% of patients preserved improvement in their functional class. These findings suggest that mavacamten not only has a short-term symptomatic effect but may also modify disease progression. (16)
In turn, aficamten has shown a similar efficacy profile. In the REDWOOD-HCM phase 2 trial, treatment with aficamten resulted in a significant reduction in the obstructive gradient of 36 mmHg, accompanied by an improvement in NYHA functional class and a decrease in NT-proBNP. (17) In the phase 3 SEQUOIA-HCM study, aficamten achieved enhanced peak VO2 of 1.9 mL/kg/min, with a favorable safety profile and a low incidence of left ventricular ejection fraction (LVEF) reduction. (18) A recently published key study is the MAPLE-HCM trial, the first to compare a myosin inhibitor with standard therapy. This phase 3 trial randomized patients with symptomatic OHCM to receive aficamten or metoprolol. At 24 weeks, aficamten achieved a significant reduction in LVOT gradient (-40.7 mmHg) in contrast to the modest reduction observed with metoprolol (-3.8 mmHg; p<0.001). Moreover, superior improvements were documented with aficamten in functional capacity (peak VO2: +1.1 vs. -1.2 ml/kg/min; p=0.001) and quality of life (KCCQ-CSS score: +15.8 vs. +8.7 points, p=0.002). In contrast, treatment with metoprolol was not associated with significant improvement in functional or symptomatic parameters. The safety profile of aficamten was favorable, with transient reductions in LVEF below 50% in 1% of patients, all of which were reversible. These findings reinforce the hypothesis that, unlike beta-blockers, myosin inhibitors such as aficamten act directly on the pathophysiological mechanism of obstruction and could redefine the initial treatment of OHCM. (19)
Table 1 summarizes the clinical trials conducted with mavacamten and aficamten.
Table 1
Main complete and published clinical trials of myosin inhibitors in adult patients with OHCM
| Study (drug) | Study type | Endpoints | Results |
|---|---|---|---|
| PIONEER-HCM (mavacamten*) | Phase II, multicenter, open label, non-randomized, n=21 Cohort A (n=11): Initial dose | Reduction of exercise LVOT gradients at 12 | Cohort A: mean change of −89.5 mmHg and 3.5 mL/kg/min |
| 2019 (12) | 10 or 15 mg Cohort B: (n=10): slow titration from 2 to 5 mg | weeks. PeakVO2 assessment | Cohort B: mean change of 25 mmHg and 1.7 mL/kg/min |
| EXPLORER-HCM (mavacamten) 2020 (13) | Phase III, multicenter, randomized, double blind, controlled with placebo, n=251 Active group n=128 Placebo n=123 | Increase of 1.5 mL/kg/ min or more in peak VO2, plus 1 point reduction in NYHA FC or Increase > 3 mL/kg/min in peak VO2 without NYHA FC worsening at 30 weeks | 37% mavacamten group vs. 17% placebo (p = 0.0005) PeakVO2: + 1.4 mL/kg/min in the mavacamten group vs. +0.1 mL/kg/min in the placebo group (p = 0.0006) Gradient: −35.6 mm Hg, p < 0.0001 |
| VALOR-HCM (mavacamten) 2022 (15) | Phase III, multicenter, randomized, double blind, controlled with placebo, n=112. | Improvement implying abandoning the indication of septal reduction at 16 week | Continue with the indication: 18% with mavacamten vs. 77% with placebo, p<0.001 Gradient: −37.2 mm Hg vs. placebo, p < 0.001 |
| REDWOOD (aficamten**) 2023 (17) | Phase II, multicenter Patients with LVOT gradients at rest ≥30 mmHg or provoked ≥50 mmHg. Assignment 2:1 aficamten/ placebo 10 weeks | Gradient changes in LVOT, LVEF and NYHA FC | -40 mmHg in the LVOT gradient at rest and - 36 mmHg in Valsalva in Cohort 1, and -43 mmHg at rest and - -53 mmHg during Valsalva in Cohort 2 |
| SEQUOIA-HCM (aficamten) 2024 (18) | Phase III, multicenter, randomized, double blind, controlled with placebo n=282 Active group n=142 Placebo n=140 | Change in peak oxygen consumption at 24 weeks Quality of life | Increase of 1.8mL/kg/min in the aficamten group vs. 0.0 mL/kg/min in placebo. ≥20 points in KCCQ-CSS: 29.7% vs. 12.4% in placebo |
| MAPLE -HCM (aficamten) 2025 (19) | Phase III, multicenter, randomized, double blind, aficamten vs metoprolol Aficamten n=87 Metoprolol n=88 | Change in oxygen consumption at 24 weeks Quality of life | Peak VO2: 1.1 mL/kg/min in aficamten and -1.2 mL in metoprolol; p=0.001 LVOT gradient: -40.7 mmHg vs. -3.8 mmHg; p<0.001 KCCQ-CSS: +15.8 vs. +8.7 points p=0.002. |
Mavacamten approved by Food Drug Administration (FDA), European Medicines Agency (EMA), and National Administration of Drugs, Food and Medical Technology (ANMAT).
** Aficamten not yet approved by FDA, EMA or ANMAT.
KCCQ-CSS: Kansas City Cardiomyopathy Questionnaire Clinical Summary Score; LVEF: left ventricular ejection fraction; LVOT: Left ventricular outflow tract; NYHA FC: New York Heart Association functional class; OHCM: obstructive hypertrophic cardiomyopathy.
Safety
In terms of safety, both drugs have been well tolerated. Between 6% and 7% of patients treated with mavacamten developed LVEF <50%, leading to temporal treatment suspension in some cases. However, this dysfunction was reversible upon drug discontinuation. In the case of aficamten, less than 2% of patients had an LVEF <50%, and in most cases ventricular function normalized after dose reduction. (13,18)
Hard events
Despite the benefits observed in terms of symptoms, oxygen consumption, and biomarkers, to date there is no clear evidence that mavacamten or aficamten reduce mortality or the incidence of ventricular arrhythmia in patients with OCHM. No reductions in mortality have been demonstrated, as clinical trials have been designed with functional and quality-of-life endpoints, without specifically evaluating long-term survival. There is also no conclusive evidence that these drugs reduce the arrhythmic burden or the risk of ventricular tachycardia and fibrillation, which are the main causes of sudden death in this population. It has been hypothesized that reducing wall stress with these agents could decrease the arrhythmic burden, but so far this has not been demonstrated in clinical trials. Regarding the need for implantable cardioverter-defibrillators, no changes in the current indication criteria have been reported.
Although studies with longer follow-up are still needed, myosin inhibitors currently represent a noninvasive alternative for the treatment of patients with OHCM who require septal intervention, such as septal myectomy and alcohol ablation. Both myectomy and septal ablation are invasive procedures associated with a very low risk of periprocedural mortality, (20,21) a risk that has not been observed with the use of myosin inhibitors.
Real-world evidence
Mavacamten has been approved by major regulatory agencies worldwide. However, to reduce the risk of left ventricular systolic dysfunction, heart failure, and drug interactions, its approval is subject to a risk evaluation and mitigation strategy (REMS) program. This program establishes education and continuous monitoring strategies, including mandatory training for healthcare professionals and periodic echocardiograms during follow-up.
In this context, Desai et al. published the results of the first 22 months of the REMS program at the end of 2024, providing data on the use of this drug in the United States. (22) Among a total of 5573 patients who received at least one dose of the drug and submitted the corresponding forms, 4.6% had a decline in LVEF, 1.3% were hospitalized for heart failure, and only 0.3% developed both complications jointly. The values were very similar in those patients who received the medication for at least one year. (22)
The drug dose used was 5 or 10 mg at six months of follow-up in most patients (74%), and only 5% required the maximum dose of 15 mg at that time. The main reason for dose reduction (95% of cases) was therapeutic adequacy, due to the achievement of a LVOT gradient < 20 mmHg. Finally, with regard to LVOT gradients, 42% persisted with resting gradients above 30 mmHg at 3 months after starting the drug, and only 29% at 6 months of treatment. As limitations, this program is designed to ensure that the conditions for safe use of the drug are met (patients with LVEF > 50% and no interactions with other drugs), without evaluating the clinical criteria for the indication, the reason for discontinuation, the evolution of symptoms, or the requirement for septal reduction therapies. The latter was evaluated in some realworld cohorts with fewer patients, also with good results. (23-25) Concerning safety strategies, the regulatory agency in Europe (EMA) requires the analysis of polymorphisms in the cytochromes responsible for its metabolism prior to treatment initiation in order to adjust the initial dose. In Argentina, the evaluation of these polymorphisms is not necessary, and monitoring depends on clinical and echocardiographic variables.
EVIDENCE IN PATIENTS WITH NOHCM
The clinical development of mavacamten in NOHCM began with the phase 2 MAVERICK-HCM study, which included symptomatic patients with LVEF ≥ 55% and no significant LVOT obstruction. At 16 weeks, a significant reduction in NT-proBNP levels was observed in the mavacamten-treated group compared with placebo, suggesting an improvement in pressure overload and diastolic function. In addition, a reduction in ventricular stiffness, measured by the E/e' index, was observed, although without significant changes in LVEF or functional capacity as measured by peak VO2. (26)
These findings led to the design of the phase 3 ODYSSEY-HCM study, which evaluated the effects of mavacamten in a population of 580 patients with symptomatic NOHCM. At 48 weeks, the drug did not meet the primary endpoint of peak VO2 improvement (+1.0 mL/kg/min with mavacamten vs. +1.3 mL/kg/ min with placebo; p=NS). However, benefits were observed in secondary parameters, including significant reductions in NT-proBNP (-50% vs. -10%; p<0.01) and ultrasensitive troponin T (-22% vs. placebo; p<0.05), as well as a favorable trend in the KCCQ-CSS score (+7 vs. +5 points). In terms of safety, a relevant finding was that 21.5% of patients treated with mavacamten developed LVEF <50%, which was reversible in most cases after treatment discontinuation.
These results suggest that, although mavacamten reduces hemodynamic load and biomarkers of myocardial stress, its effect on overall functional capacity is limited. One possible explanation is that the dose used could have been excessive and the exposure time relatively short, which could have attenuated the net clinical benefit observed in this population. (27)
In the case of aficamten, the phase 2 FORESTHCM trial has shown encouraging preliminary results, with a reduction in NTproBNP and improved echocardiographic parameters of diastolic dysfunction. (28) The phase 3 ACACIA-HCM study is currently underway, evaluating the efficacy and safety of aficamten in a large cohort of patients with NOHCM, and will provide key evidence on its impact on cardiac function and symptoms.
MEDICATION TITRATION
Mavacamten is administered orally in 2.5 mg, 5 mg, 10 mg, and 15 mg capsules. The method of use, dosage, and titration are based on the regimen used in the EXPLORER-HCM study. (13)
The recommended starting dose is 5 mg once daily. Dose adjustment is suggested every 4 weeks, with a maximum daily dose of 15 mg. Strict clinical monitoring is essential to identify possible signs of heart failure, supplemented by echocardiographic monitoring focused mainly on the evaluation of LVEF and LVOT gradient, both before the start of treatment and during the subsequent follow-up. Treatment initiation is not recommended in patients with LVEF < 50%. Once initiated, it is advised to follow the scheme in Figure 2. (adapted from 29)
Fig. 2
Mavacamtem titration algorithm. Adapted from (29)
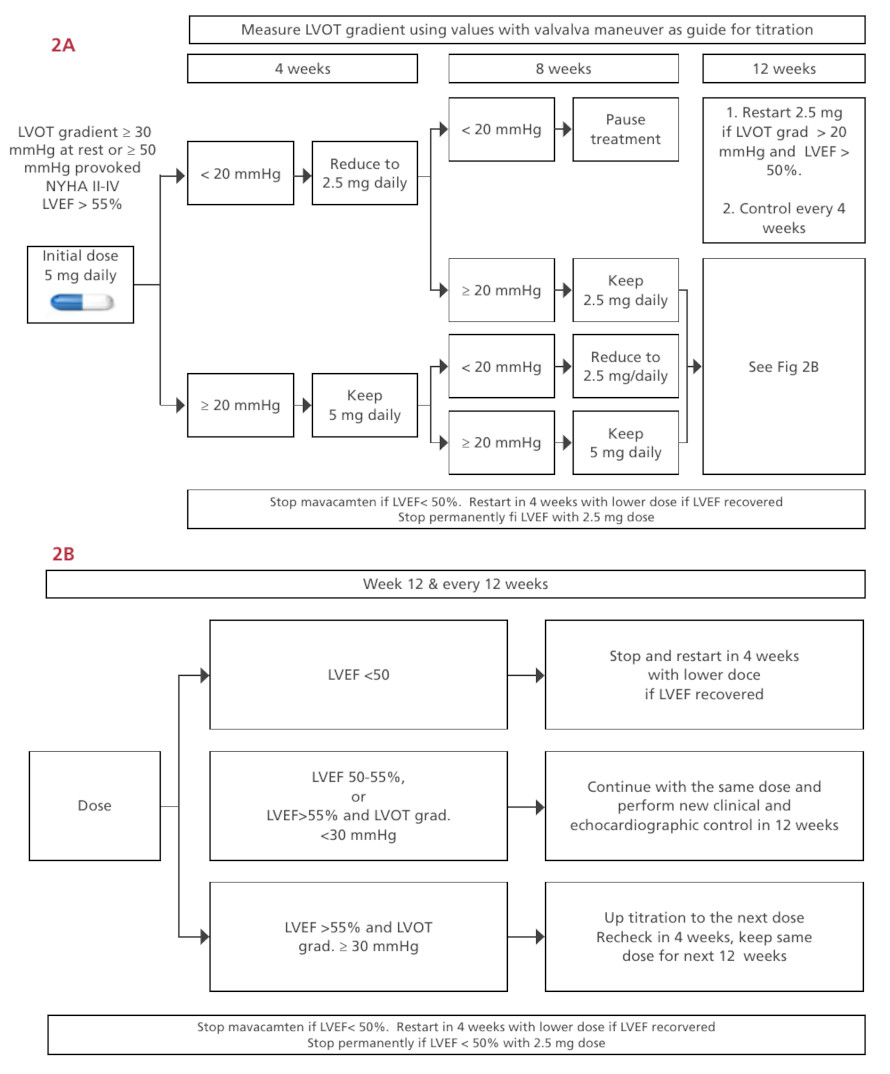
LVEF: left ventricular ejec tion fraction; grad: gradient; LVOT: left ven tricular outflow tract
In the case of aficamten, the initial dose is 5 mg once daily with adjustments every 2 weeks due to its shorter half-life. Figure 3 outlines the titration algorithm used in the phase III SEQUOIA-HCM study. (18)
Fig. 3
Titration of aficamten in SEQUOIA-HCM and MAPLE trials
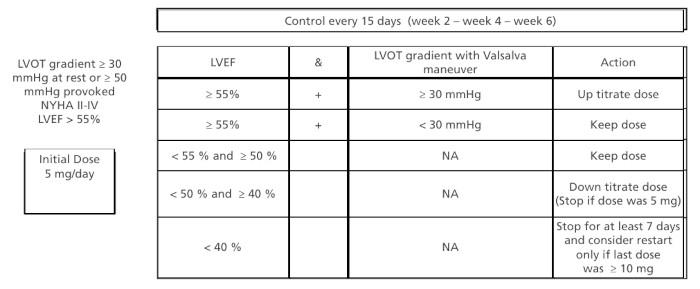
LVEF: left ventricular ejection fraction; LVOT: Left ventricular out flow tract; NA: not available; NYHA FC: New York Heart Association functional class
Clinically relevant and frequent drug interactions
Given its metabolism through the cytochrome system (mainly CYP2C19), the use of mavacamten should be avoided with drugs such as certain antifungals, certain macrolide antibiotics, some antidepressants, and protease inhibitors, among others, as well as grapefruit juice. CYP2C19 inhibitors and strong CYP3A4 inhibitors should be discontinued for at least 14 days before starting mavacamten. Aficamten does not interact with the cytochrome system, so there would be no such interaction and no further precautions would be necessary. Table 2 summarizes some of the relevant pharmacologic interactions for mavacamten.
Table 2
Relevant pharmacological interactions for mavacamten
| Group | Effect | Example | Recomendation |
|---|---|---|---|
| Strong 2C19 inhibitors | Increase mavacamten plasma concentration | Fluoxetine, modafinil, ritonavir | Do not combine |
| Strong 3A4 inhibitors | Increase mavacamten plasma concentration | Clarithromycin, itraconazol, ritonavir, lopinavir, loperamide, efavirenz | Do not combine |
| Moderate 2C19 inhibitors | Slightly increase mavacamten plasma concentration | Sertraline, efavirenz, pantopra- zole, lansoprazole, omeprazole, clarithromycin | Lower dose to 2.5 mg when starting any of these drugs |
| Moderate 3A4 inhibitors | Slightly increase mavacamten plasma concentration | Erythromycin, fluconazole, amiodarone, diltiazem, verapamil, grapefruit juice | Lower dose to 2.5 mg when starting any of these drugs |
| 3A4 and 2C19 inducers | Decrease mavacamtem con- centration and increase it when discontinued | Rifampicin, phenytoin, dexameth- asone, modafinil, efavirenz | Precaution when suspending any of these drugs |
COST-EFFECTIVENESS
The high cost of mavacamten raises questions about its cost-effectiveness compared with other therapeutic strategies. While it offers significant clinical benefits, its current price could place it outside the generally cost-effectiveness threshold accepted in some healthcare systems.
An analysis conducted in the United States by the Institute for Clinical and Economic Review (ICER) established that, for mavacamten to be considered cost-effective according to the thresholds used in that country, its annual price would need to be less than $15 000 (far from the current $85 000). (30) However, these conclusions are not directly applicable to other health systems, as the costs of treatments and procedures vary considerably between countries.
While mavacamten could reduce the need for invasive procedures and improve patients' quality of life, the cost remains a challenge for its widespread implementation. Its economic viability will depend on access strategies, price negotiations, and additional studies to assess its impact in real-world clinical practice.
As for aficamten, it is not yet commercially available, so it is not possible to analyze its cost-effectiveness at this moment.
CONCLUSION
Myosin inhibitors have proven to be a safe and effective option in the treatment of OCHD, currently positioning them as a second-line alternative. However, more evidence is still needed to consider them as a first-line treatment, especially in terms of their structural impact and their long-term effect on hard clinical events, such as mortality, arrhythmias, and need for devices. Their potential to modulate disease progression is a key aspect to investigate, particularly in relation to the regression of hypertrophy and the reduction of myocardial fibrosis.
On the other hand, their high cost and difficulty of access in many healthcare systems, including ours, limit their availability and underscore the importance of careful selection of patients who can benefit most. Despite these challenges, their use in nonobstructive forms and in asymptomatic patients is promising, opening new perspectives in the management and possible modification of the natural course of HCM.
Conflicts of interest
None declared.
(See authors' conflict of interests forms on the web/Additional material).
